- PATHOLOGY / SYMPTOMS:
- Urethral cancer
- Penile cancer
- Testicular cancer
- Bladder cancer
- Prostate cancer
- Kidney cancer
- Upper urinary tract cancer
- Benign tumors of the adrenal gland
- Malignant tumor of the adrenal gland
- Pathology of the urachus
- TREATMENTS:
- URETHRAL CANCER.
- PENILE CANCER.
- TESTICULAR CANCER.
- BLADDER CANCER.
- PROSTATE CANCER.
- KIDNEY CANCER.
- UPPER URINARY TRACT CANCER.
- BENIGN TUMORS OF THE ADRENAL GLANDS.
- MALIGNANT TUMOR OF THE ADRENAL GLANDS (ADRENAL CARCINOMA).
- PATHOLOGY OF URACHUS.
Urethral cancer
Urethral cancer is a disease that involves the formation of cancerous or malignant cells in the tissues that comprise the urethra, the tube that carries urine from the bladder to the outside of the body. The size of this varies according to sex, in men it has a larger size than in women.
This type of cancer is rare in the population and there are more cases in men.
Aetiology
The causes of urethral cancer are unclear. There are people who have more risk factors for this disease, although this does not mean that you will get it. Factors that can increase the chances are:
- Have a family history with this pathology.
- Having a condition that affects the urethra, causing chronic inflammation. The most important are: sexually transmitted infections, such as human papillomavirus (HPV) and recurrent urinary tract infections.
- Having a history of bladder cancer, since implants may appear in the urethra during the evolution of this disease.
Symptoms
The signs of this disease are not usually shown in the early stages. Among the most frequent we find:
- Difficulty urinating.
- Frequent urination, especially at night.
- Weak or interrupted urination flow, often coupled with difficulty emptying the bladder completely.
- Incontinence
or inability to control urine in the bladder. - Discharge from the urethra.
- Presence of blood in the urine.
- Thickening of the penis in men or the presence of a lump in women.
Subtypes
Variants of urethral cancer depend on the tissues lining this duct and the types of cells that make it up. The pathology can spread and metastasize to nearby tissues or lymph nodes. Known subtypes of urethral cancer are:
- Squamous cell carcinoma is the most common type of urethral cancer.
- Transitional cell carcinoma forms in the area near the urethral opening in women and in the part of the urethra that passes through the prostate in men.
- Adenocarcinoma forms in the glands surrounding the urethra in men and women.
Depending on the location of the tumor, we can differentiate between:
- Distal urethral cancer: in the area closest to the outside of the body.
- Proximal urethral cancer: located in the part closest to the bladder.
Diagnosis
In the diagnosis of urethral cancer, multiple tools are used, since it must be determined not only the presence of tumor implants in the organ, but also the possibility that it has spread to other parts of the body. In the diagnosis, information is collected through:
- Physical exam and health history: An exam of the body to check overall health and identify any signs of disease, such as lumps or anything else that seems abnormal. Data on health habits, history of illnesses, and previous treatments are also taken.
- Pelvic exam: Examination of the vagina, cervix, uterus, fallopian tubes, ovaries, and rectum.
- Digital rectal exam: rectal exam
- Urine cytology: A laboratory test in which urine is viewed under a microscope to look for abnormal cells.
- Urinalysis: A test to analyze the color of urine and its contents.
- Blood tests: Tests in which a blood sample is examined to measure the amount of certain substances released into the blood by organs and tissues in the body.
- Urethroscopy: A procedure to look inside the urethra and check for abnormal areas.
- Biopsy: removal of cells or tissue samples from the urethra.
The process used to find out if cancer has spread within the urethra or to other parts of the body is called staging. The information obtained in the staging process determines the stage of the disease. It is important to know what stage the disease is at in order to plan your treatment.
The following procedures are used during the staging process:
- Chest x-ray: X-ray of the organs and bones inside the chest.
- Computed tomography (CT) scan of the pelvis and abdomen: A procedure that makes a series of detailed pictures of the inside of the body from different angles.
- Magnetic resonance imaging (MRI)
- Urethrogram: A series of x-rays of the urethra. Contrast is injected through the urethra into the bladder.
Treatments
There are different types of treatment available for urethral cancer patients.
Treatments:
Surgery
Surgery to remove the cancer is the most common treatment for urethral cancer. Sometimes one of the following types of surgery is done.
- Open resection: removal of the cancer by surgery.
- Transurethral resection (TUR): Surgery to remove the cancer using a special tool inserted into the urethra.
- Electroresection with fulguration: Surgery to remove cancer using an electric current.
- Laser surgery: A surgical procedure in which a laser beam is used like a scalpel to make bleeding-free cuts in the tissue or to remove or destroy tissue.
- Lymph node dissection: Sometimes lymph nodes in the pelvis and groin are removed.
- Cystouretrectomy: Surgery to remove the bladder and urethra.
- Cysthoprostatectomy: Surgery to remove the bladder and prostate.
- Anterior pelvic exenteration: Surgery to remove the urethra, bladder, and vagina.
- Partial penectomy: Surgery to remove the part of the penis that surrounds the urethra to where the cancer has spread.
- Radical penectomy: Surgery to remove the entire penis.
If the urethra is removed, the surgeon will open a new avenue for urine to leave the body. This procedure is called urine shunting. The surgeon sometimes uses a part of the small intestine to make a tube for urine to pass through an opening (stoma). This is called an ostomy or urostomy.
Once the doctor removes all of the cancer visible at the time of surgery, some patients may receive chemotherapy or radiation therapy after surgery to kill any remaining cancer cells. Treatment given after surgery to decrease the risk of the cancer coming back is called adjuvant therapy.
Radiotherapy
Radiation therapy is a cancer treatment that uses high-energy x-rays or other types of radiation to kill cancer cells or stop them from growing.
There are two types of radiation therapy:
- External radiation therapy: A type of radiation therapy that uses a machine that sends radiation to the area with cancer from outside the body.
- Internal radiation therapy: A type of radiation therapy that uses a radioactive substance sealed in needles, seeds, wires, or catheters that are placed directly into or near the cancer. Internal radiation therapy is also called brachytherapy.
The way radiation therapy is given depends on the type of cancer and where in the urethra the cancer formed. External and internal radiation therapy are used to treat urethral cancer.
Chemotherapy
Chemotherapy is a cancer treatment that uses drugs to stop cancer cells from growing, either by killing them or by stopping them from dividing.
Depending on the location of the cancer, certain treatments may be used and others may not.
For distal urethral cancer…
Treatment of distal urethral cancer is different for men and women.
For women, treatment may include the following:
- Surgery to remove the tumor (transurethral resection), electroresection and fulguration, or laser surgery for tumors that have not spread deep into the tissue.
- Brachytherapy (internal radiation therapy) or external radiation therapy for tumors that have not spread deep into the tissue.
- Surgery to remove the tumor (anterior pelvic exenteration) for tumors that have spread deep into the tissue. Nearby lymph nodes are also sometimes removed (lymph node dissection). Radiation therapy is sometimes given before surgery.
For men, treatment may include the following:
- Surgery to remove the tumor (transurethral resection) electroresection and fulguration, or laser surgery for tumors that have not spread deeply into the tissue.
- Surgery to remove part of the penis (partial penectomy) for tumors that are near the tip of the penis. Nearby lymph nodes are also sometimes removed (lymph node dissection).
- Surgery to remove part of the urethra for tumors that are in the distal urethra but are not at the tip of the penis or spread deep into the tissue. Nearby lymph nodes are also sometimes removed (lymph node dissection).
- Surgery to remove the penis (radical penectomy) for tumors that have spread deep into the tissue. Nearby lymph nodes are also sometimes removed (lymph node dissection).
- Radiation therapy, with or without chemotherapy.
- Chemotherapy given along with radiation therapy.
For proximal urethral cancer…
Treatment for proximal urethral cancer or urethral cancer that affects the entire urethra is different for men and women.
For women, treatment may include the following:
- Radiation therapy or surgery (open resection, transurethral resection) for tumors that are 3/4 inch or smaller.
- Radiation therapy followed by surgery (anterior pelvic exenteration with lymph node dissection and urine diversion).
For men, treatment includes the following:
- Radiation therapy or radiation therapy and chemotherapy, followed by surgery (cystoprostatectomy, penectomy, lymph node dissection, and urine diversion).
For urethral cancer resulting from bladder cancer
Treatment of urethral cancer that forms at the same time as invasive bladder cancer may include the following:
- Surgery (cystoretrectomy for women or urethrectomy and cystoprostatectomy for men).
For metastatic urethral cancer…
Treatment for urethral cancer that metastasized (spread to other parts of the body) is usually chemotherapy.
Treatment of recurrent urethral cancer may include one or more of the following:
- Surgery to remove the tumor. Nearby lymph nodes are also sometimes removed (lymph node dissection).
- Radiation therapy.
Penile cancer
Risk factors
Human papillomavirus (HPV) infection is the risk factor most associated with penile cancer. There are more than 100 serotypes, but only some increase the possibility of cancer in the genital organs.
Other factors involved are:
- Age over 60 years.
- Phimosis (condition in which the foreskin of the penis cannot move backwards over the glans).
- Inadequate personal hygiene.
- Consumption of tobacco products.
Symptoms
Signs and symptoms that suggest penile cancer include:
- Redness, irritation, or a sore on the penis.
- Ulcers that do not heal.
- A mass on the penis.
Diagnosis
The following is done:
- Physical exam and history: Examination of the body to check general health and identify any signs of penile disease, such as lumps or anything else that seems abnormal. Data on health habits, history of illnesses, and previous treatments are also taken.
- Biopsy: The removal of cells or tissues for a pathologist to look at under a microscope and check for signs of cancer. The tissue sample is removed using one of the following procedures.
- Incisional biopsy: The removal of a part of a mass or sample of tissue that does not look normal.
- Excisional biopsy: The complete removal of a mass or area of tissue that does not look normal.
Staging
In diagnosis it is important to determine if penile cancer is localized or has spread to other regions of the body. Several complementary tests are used to place the case within a specific clinical stage: 0, I, II, III or IV. It is important to know the stage to plan treatment.
Stages of Penile Cancer
The different stages are:
Stage 0
Stage 0 is divided into stages 0is and 0a.
- In stage 0is, abnormal cells are found on the surface of the skin of the penis. These abnormal cells form lumps that sometimes become cancerous and spread to
- In stage 0a, squamous cell cancer that does not spread is found on the surface of the skin of the penis or on the inner surface of the foreskin of the penis. Stage 0a is also called localized noninvasive squamous cell carcinoma.
Stage I
In stage I, cancer has formed and spread to tissue just under the skin of the penis. Cancer has not spread to the lymphatic vessels, blood vessels, or nerves. Cancer cells look like normal cells when viewed under a microscope.
Stage II
Stage II is divided into stages IIA and IIB.
In stage IIA, cancer has spread as follows:
- to the tissue just under the skin of the penis. Cancer has spread to lymphatic vessels, blood vessels, or nerves; or to the tissue just under the skin of the penis. Cancer cells look very abnormal or are sarcomatoid when viewed under a microscope; or
- to the corpus spongiosum (erectile spongy tissue of the shaft of the penis and glans that fills with blood during an erection).
In stage IIB, cancer
- through the connective tissue layer surrounding the corpus cavernosum (erectile spongy tissue of the shaft of the penis) and into the corpus cavernosum.
Stage III
Stage III is divided into stages IIIA and IIIB. Cancer is found in the penis.
- In stage IIIA, cancer has spread to 1 or 2 lymph nodes on one side of the groin.
- In stage IIIB, cancer has spread to 3 or more lymph nodes on one side of the groin, or to lymph nodes on both sides of the groin.
Stage IV
In stage IV, cancer has spread as follows:
- to tissues near the penis, such as the scrotum, prostate, or pubis, and may have spread to lymph nodes in the groin or pelvis ; or
- to one or more lymph nodes in the pelvis, or cancer has spread through the outer covering of lymph nodes to nearby tissue; or
- lymph nodes outside the pelvis or to other parts of the body, such as the lungs, liver, or bones.
Treatments
Surgery
Surgery is the most common treatment for all stages of penile cancer. A doctor may remove the cancer using one of the following operations:
- Mohs micrographic surgery: A procedure in which the skin tumor is cut into thin layers. During surgery, the edges of the tumor and each layer of the tumor removed are looked at under a microscope to check for cancer cells. Layers are removed until cancer cells are no longer visible. This type of surgery removes as little normal tissue as possible and is often used to remove cancer that is in the skin. This procedure is also called Mohs surgery.
- Laser surgery: A surgical procedure in which a laser beam is used like a scalpel to make bleedingless cuts in tissue or to remove a superficial lesion, such as a tumor.
- Cryosurgery: Treatment that uses an instrument to freeze and destroy abnormal tissue. This type of treatment is also called cryotherapy.
- Circumcision: Surgery to remove part or all of the foreskin of the penis.
- Wide local excision: Surgery to remove cancer and some of the healthy tissue around it.
- Penile amputation: Surgery to remove part or all of the penis. If part of the penis is removed, amputation is a partial penectomy. If the entire penis is removed, it is a total penectomy.
During surgery, lymph nodes in the groin are sometimes removed.
After the doctor removes all of the cancer visible at the time of surgery, some patients may receive chemotherapy or radiation therapy to kill any remaining cancer cells. Treatment given after surgery to decrease the risk of the cancer coming back is called adjuvant therapy.
Radiotherapy
Radiation therapy is a cancer treatment that uses high-energy x-rays or other types of radiation to kill cancer cells or stop them from growing. There are two types of radiation therapy.
- External radiation therapy: A type of radiation therapy that uses a machine that sends radiation to the cancer from outside the body.
- Internal radiation therapy: A type of radiation therapy that uses a radioactive substance sealed in needles, seeds, wires, or catheters that are placed directly into or near the cancer.
Chemotherapy
Chemotherapy is a cancer treatment that uses drugs to stop cancer cells from growing, either by killing them or by stopping them from dividing.
Biologic drugs
Biologic therapy is a treatment in which the patient’s immune system is used to fight cancer. Substances made by the body or produced in a laboratory are used to boost, direct, or restore the body’s natural defenses against cancer. This type of cancer treatment is also called biotherapy or immunotherapy. Topical biologic therapy with imiquimod is sometimes used to treat stage 0 penile cancer.
Sentinel node biopsy
Sentinel lymph node biopsy is the removal of the sentinel lymph node during surgery. The sentinel lymph node is the first node in a group of lymph nodes to receive lymphatic drainage from the primary tumor. It is the first lymph node where cancer from the primary tumor is likely to spread. A radioactive substance or blue dye is injected near the tumor. The substance or dye flows through the lymph ducts to the lymph nodes. The first node that receives the substance or dye is removed.
A pathologist looks at the tissue under a microscope to look for cancer cells. If no cancer cells are found, no more lymph nodes may need to be removed. Sometimes a sentinel lymph node is found in more than one group of lymph nodes. After sentinel lymph node biopsy, the surgeon removes the cancer.
Depending on each stage, certain types of treatments may be used and others may not.
Stage 0
Treatment for stage 0 may include one of the following:
- Mohs microsurgery.
- Topical chemotherapy.
- Topical biological therapy with imiquimod.
- Laser surgery.
- Cryosurgery.
Stage I
If the cancer is found only in the foreskin, only wide local excision and circumcision treatment may be needed.
Treatment for stage I penile cancer may include the following:
- Surgery (partial or total penectomy, with or without removal of lymph nodes in the groin).
- External or internal radiation therapy.
- Mohs microsurgery.
- A clinical trialof laser therapy.
Stage II
Treatment for stage II penile cancer may include the following:
- Surgery (partial or total penectomy, with or without removal of lymph nodes in the groin).
- External or internal radiation therapy followed by surgery.
- A clinical trial of sentinel lymph node biopsy followed by surgery.
- A clinical trial of laser surgery.
Stage III
Treatment for stage III penile cancer may include the following:
- Surgery (penectomy and removal of lymph nodes in the groin, with or without radiation therapy).
- Radiotherapy.
- A clinical trial of sentinel lymph node biopsy followed by surgery.
- A clinical trial of radiosensitizers.
- A clinical trial of chemotherapy before or after surgery.
- A clinical trial with new drugs, biological therapy, or new types of surgery.
Stage IV
Treatment for stage IV penile cancer is usually palliative (to relieve symptoms and improve quality of life). Treatment may include the following:
- Surgery (wide local excision and removal of lymph nodes in the groin).
- Radiation therapy.
- A clinical trial of chemotherapy before or after surgery.
- A clinical trial with new drugs, biological therapy, or new types of surgery.
Testicular cancer
Testicular cancer is the most common tumor in men between the ages of 15 and 35. However, after this peak of incidence it becomes a rarity and, in general, only accounts for 1% of all tumors diagnosed in men. In Spain, approximately 1,300 new cases were diagnosed in 2019. Its frequency varies in different countries for reasons that we do not know.
Since the 90s the number of diagnosed cases is increasing for unknown reasons, although mortality has decreased, reflecting improvements in the treatment of these patients.
Fortunately, the vast majority of patients are cured, even in those cases in which the disease initially presents already very advanced. This is possible mainly thanks to chemotherapy regimens.
Aetiology
The specific cause of testicular cancer is unknown, as are many other tumors; But some risk factors with a clear association with germline tumors have been identified.
The main risk factor is cryptorchidism (lack of descent from the testicle to the scrotum during the first years of life).
Between 1 and 3% of patients with germline tumors have a family history of testicular cancer, suggesting a hereditary component. In the same way, the siblings or children of these patients have up to 10 times more risk of developing the disease. However, the genetic factors responsible are not yet well established.
Prevention
Generally speaking, there is no way to prevent testicular cancer, because there are no known risk factors that can be avoided. In addition, the frequency of the disease is low, even in people who have risk factors. On the other hand, it has been shown that orchidopexy (intervention by which an undescended testicle is lowered and fixed to the scrotum naturally) prevents the development of testicular cancer in the subgroup of boys with cryptorchidism.
It is not recommended to perform routine examinations or tests to diagnose it early in the general population, since it is a rare tumor, easy to diagnose in early stages and with a good prognosis. However, it is important that the target population of this disease (men between 15-35 years) is familiar with the most common warning signs and symptoms.
Symptomatology
The vast majority of male germ tumors are diagnosed because the patient feels a lump in the testicle, which often does not hurt. In cases where there is pain, it usually appears progressively, although it can do so suddenly if the tumor is complicated by an infection (orchiepididymitis) or if it bleeds. In general, patients describe more a feeling of weight in the genital area or simply an increase in the size of the testicle.
In much rarer cases, these tumors can cause breast growth in males (gynecomastia).) by increased chorionic beta-gonadotropin (β-HCG). In some patients it may exist Back or abdominal pain, caused by painful stimuli from the testicle being directed to that site, or by metastases (extensions of the testicular tumor to other areas) in lymph nodes in the back of the abdomen (retroperitoneum).
Finally, in cases where there are metastases in other organs (for example, in the lung, brain, bone, etc.), these can produce very varied symptoms (shortness of breath, instability, pain, etc.), depending on their location.
Histological types
Histologic types are the different kinds of tumors that can develop in an organ. These types are determined based on the microscopic characteristics of the tumor.
In the case of testicular tumors, when the germ cell grows abnormally, it can follow different patterns and give rise to two large families of tumors:
– Seminomas: these represent approximately 50% of germline tumors and usually appear on average 10 years later (around the fourth decade of life). For a testicular tumor to be considered seminoma, it must not be mixed with other types of tumor. There are two subtypes:
- Classic seminoma
- Atypical seminoma
– Nonseminomas: nonseminoma tumors usually appear during the third decade of life. When we talk about “non-seminome” we are actually referring to a group that includes several possible subtypes:
- Embryonal carcinoma
- Choriocarcinoma
- Endodermal sinus tumor
- Teratoma
The most common subtype is embryonal carcinoma, although patients usually have mixed tumors that mix in different proportions any of these subtypes and even parts of seminoma (in this particular case, they continue to be considered nonseminomas).
Diagnosis
When a probable case of testicular cancer is addressed, in addition to asking about the symptoms and exploring the patient, the study is completed with the following complementary tests:
- General Analytics, including LDH, alpha-fetoprotein, and β-hCG. These substances are called Tumor markers. They are proteins produced by tumor cells and will be very useful for the diagnosis and / or monitoring of the disease. β-HCG may be elevated in any type of testicular tumor, while alpha-fetoprotein only increases in nonseminoma tumors. LDH (lactate dehydrogenase) is another protein in the blood that is also elevated in some patients with testicular tumors, and its evolution should be monitored during treatment. However, as with other types of cancer, tumor markers do not increase in all patients with testicular cancer; nor does any elevation of these proteins correspond unequivocally with the fact of having a germ tumor.
- Ultrasound of the testicle: It is a crucial test, since it allows us to distinguish a tumor from other benign entities that could also cause a palpable nodule in the testicle. If the existence of a tumor is definitively confirmed, ultrasound can observe its characteristics (location, size, vascularization, etc.), explore the contralateral testicle, etc.
- Computed axial tomography (CT) of the chest, abdomen and pelvis: it allows to study the rest of the organs of the body and the main routes of testicular lymphatic drainage, so that it is essential for a correct staging of the case.
In addition, they can be performed in some cases:
- CT or brain magnetic resonance imaging (MRI): if brain metastases are suspected.
- Bone scan: if bone metastases are suspected.
- Positron emission tomography (PET): sometimes used to assess tumor remnants that may persist after initial chemotherapy treatment in cases of advanced disease. PET is not indicated as a routine procedure in the initial diagnosis of all cases.
Staging
- Stage I: The tumor only affects the testicle.
- Stage II: The tumor spreads to lymph nodes, usually located in the retroperitoneum (the area behind the abdomen).
- Stage III: The tumor has metastasis or very significant elevation of tumor markers.
Prognosis
The prognostic factors are characteristics of the tumor and the patient that allow to estimate a priori its subsequent evolution, complementing the stage marked by the TNM system.
In testicular cancer with metastasis treated with chemotherapy there are 3 prognostic factors of great importance with which in 1997 a prognostic classification of enormous scope was developed, still in force, and critical when determining the most appropriate treatment for each patient: The International Germ Cell Cancer Collaborative Group (IGCCCG) Prognostic Classification.
This classification establishes 3 categories for nonseminoma tumors (favorable, intermediate or unfavorable prognosis); and 2 for seminomatous (favorable or intermediate prognosis; there are no patients with an unfavorable prognosis in this histological subtype). Prognostic factors are:
- The tumor originates in the testicle itself (a sign of good prognosis) or outside it. A small percentage of germ cell tumors may occur in areas of the body other than the testicle, with no tumor found in the testicle itself; either because the initial tumor has disappeared or because the disease actually originated outside the testicle. The latter can happen because during the development of the embryo the testicle migrates to its final position in the scrotum. In this journey there may be remains of testicular cells that very rarely already in adulthood can give rise to tumors.
- The absence of distant metastases (M1) (sign of good prognosis), with the exception of lung metastases that alone do not indicate a poor prognosis.
- The degree of elevation of tumor markers. In general, the higher, the worse the prognosis.
Treatment
Although germline tumors of the testicle have a very good prognosis in experienced hands, we are talking about very fast-growing neoplasms, which represent a very important aggression for the body, and that usually affect very young patients. That is why the main international recommendation is that these patients be quickly referred to specialized centers with professionals accustomed to their management.
It must be borne in mind that, once cured, these patients will have been subjected to treatments that reduce their reproductive capacity, and that impact on their sexual sphere and self-image, so we must foresee these details from the beginning, involve their partners if any, preserve sperm if appropriate, and advise properly at the right time.
Treatment of testicular tumors first requires removing the testicle through a surgical procedure called orchiectomy. This is one of the few tumors in which, even if the disease has spread to other organs, the original tumor must be removed. The reasons are that chemotherapy treatments get poorly to the testicle and that the examination of the tumor is very important to determine its characteristics and treatment. Surgery to remove the testicle should always be performed through the groin (groin line) and not through the skin of the scrotum (transscrotal), otherwise the chances of contamination of healthy tissue by tumor cells increase.
Once the testicle is removed, treatment is decided based on:
- Tumor type (seminoma or nonseminoma): Treatment of seminomas and stage I and II nonseminoma tumors is different, but in stage III it is relatively similar;
- Of its extension (stage I, II or III); and
- Of its prognostic category (favorable, intermediate, or unfavorable).
Although here the most common treatments are briefly discussed.
Treatment according to the different stages
- Stage I:
Patients with stage I testicular tumors are cured with proper treatment in nearly 100% of cases. As previously mentioned, these tumors only affect the testicle, so in theory their removal should be curative by itself. The problem is that in a small percentage of patients the disease can recur. To avoid this, in some cases complementary treatment can be administered with preventive intention (adjuvant treatment) that significantly reduces the risk of the disease returning. Adjuvant treatments (both chemotherapy and radiotherapy) involve risks and can generate sequelae.
- Seminoma:
80% of patients are cured after orchiectomy, but in 20% of cases, the disease may recur. Therefore, in patients with stage I seminoma, the alternatives are:
– Give chemotherapy with 1 or 2 cycles of a drug called carboplatin. This treatment is well tolerated and decreases the risk of relapse to 3-4%.
– Do not administer any treatment and make careful observation of the patient. This avoids unnecessary treatments for patients who were not really going to have a relapse.
– Treatment with retroperitoneal radiotherapy. These tumors are very radiosensitive and radiation therapy is effective in treating them. However, today this therapeutic option is less used by late toxicities.
- Non-seminome:
In nonseminoma tumors, 70% of patients are cured with surgery alone, but in the remaining 30% the tumor may recur. Nowadays we do not have factors that segregate us well which patients are going to relapse and which are not.
In principle, those patients whose tumor invades the blood vessels or lymphatic vessels of the testicle are more at risk of the disease returning. In this case it may be more justified to treat preventively with chemotherapy by administering two cycles with drugs called bleomycin, etoposide and cisplatin (BEP). However, with this strategy we would be overtreating a percentage of patients close to 50% who will never relapse after being operated.
Patients whose tumor does not invade the blood or lymphatic vessels have less risk of the disease coming back and tend to be less aggressive and generally tend to follow only carefully.
Another option is to monitor all patients and treat only those in whom the disease recurs to avoid unnecessary treatments, as discussed above.
Also in non-seminoma tumors, in case the disease reappears, whether complementary chemotherapy has been administered or not, the treatment is adapted to the advanced tumor scenario.
- Stage II:
Patients with stage II have disease in the nodes of the retroperitoneum. Most of these patients are also cured with proper treatment.
- Seminoma:
the usual treatment of these patients consists of chemotherapy with 3 cycles of BEP or four cycles of cisplatin and etoposide (EP), which achieve an equivalent result. In some patients, remnants of the lymph nodes in the retroperitoneum remain after these treatments that do not disappear completely. In this situation there are two options depending on the size of the residual mass (remains of the tumor). In those residual masses greater than 3 cm, a PET scan should be considered to confirm or deny if there is viable disease or it is only necrosis (dead cells) or fibrosis. In residual lesions smaller than 3 cm, no intervention is necessary and it is enough to follow the patient routinely.
- Nonseminoma:
In these patients, the most common treatment is to administer chemotherapy with 3 cycles of BEP. Also in these patients there may be remnants of the lymph nodes of the retroperitoneum that do not disappear completely. In these cases, any residual > lesion 1 cm must be resected and analyzed by an intervention called retroperitoneal lymphadenectomy, which can be carried out robotically, laparoscopically or openly. In this intervention, the lymph nodes of the entire retroperitoneal space are resected, that is, the nodes located around the great vessels of the body (presacral space, obturator space, iliacs, aorta, cava, and renal vessels). PET should not be used as a diagnostic method in this context except in very exceptional situations.
- Stage III: In these patients, treatment is performed in a similar way in both nonseminoma tumors and seminomas. These patients should be classified according to the risk criteria they present and treated according to the risk group to which they belong.
- Patients with a favorable prognosis:
treatment consists of 3 cycles of BEP or 4 cycles of PE.
- Patients with intermediate or poor prognosis: treatment consists of 4 cycles of BEP. In some patients, alternatives such as different chemotherapy regimens or high-dose chemotherapy with bone marrow progenitor support, are offered, occasionally in the context of clinical trials, with the aim of trying to improve treatment outcomes.
Monitoring and controls
After treatment for testicular cancer, regular check-ups are very important. Screening is more intense in stage I patients who are not given adjunctive therapy after surgery because their risk of recurrence is higher. In these patients, it is common for check-ups to be performed every 2 months, or even every month, during the first and second year of diagnosis.
At check-ups, in addition to asking about symptoms and performing a physical examination, a determination of tumor markers of testicular tumors is usually performed. In addition, chest x-ray, abdominal CT and testicle ultrasound are usually performed periodically. Depending on the extent of the disease to the diagnosis and the symptoms presented by the patient, other different tests may be added.
The frequency of reviews is often variable and their duration is a matter of debate.
The reality is that the greatest risk of the disease coming back occurs in the first 2 years after diagnosis. After 5 years [que hoy en día se considera un punto de referencia muy común en oncología] the risk of relapse is low.
Bladder cancer
Bladder cancer is a much more frequent tumor than what statistics or the media sometimes transmit to us. It ranks ninth in terms of the number of cancer diagnoses globally, and fifth in Europe.
The average age at diagnosis is around 70 years. Curiously, there are marked geographical differences, with tumors of the urinary tract being much more frequent in Western Europe (including Spain) and North America, than in Asia or Eastern Europe.
Most of these tumors are diagnosed in early stages of development and can be cured with more or less extensive surgeries. However, when the tumor is more advanced, cure can be more complicated and the goals of treatment change to prolong survival and improve the patient’s quality of life.
Risk factors
The main known cause of bladder cancer is tobacco, which accounts for around 50% of all diagnosed cases. Tobacco doesn’t just affect our airway through smoke. And not only consumption, but environmental exposure to tobacco can also be considered as a risk factor. More than 60 carcinogenic products contained in cigarettes are absorbed and eliminated in the urine greatly affecting the cells of the wall of our urinary tract.
There are other risk factors, but most of them are not common in our environment. These factors include exposure to some industrial chemicals (certain metals, dyes, and gums); exposure to drugs such as cyclophosphamide, or schistosomiasis, an infection caused by a parasite that is usually found in Africa and certain regions of Latin America.
It has not been clearly shown that the presence of a family history of bladder cancer increases the risk of developing the disease in other members of the same family, although diagnosis in patients younger than 60 years should be considered with special care.
Prevention
The best way to prevent bladder cancer is to avoid tobacco use, since as we have said it is the most important risk factor for developing the disease.
Healthy people are not routinely screened to diagnose bladder cancer early, because there is no evidence that this is actually beneficial.
Histological types
Histologic types are the different kinds of tumors that can develop in an organ, determined based on the microscopic characteristics of the tumor.
In the case of bladder tumors, the main histological type is urothelial carcinoma, whose cells derive from the mucosa that lines the bladder inside (transitional epithelium or urothelium). As in other organs, tumors derived from many other cells can occur, including squamous cell carcinoma (which forms on flat cells that appear in the bladder when there is irritation of the bladder), adenocarcinoma (tumors that form from glands), small cell carcinomas and others even less frequent. In this document we will refer to urothelial tumors, as they are by far the most common.
Tumors derived from the upper urinary tract (ureter and renal pelvis) are also usually urothelial. They are very similar in their biological behavior to bladder cancer and their management, except for specific technical aspects, is very similar.
Symptomatology
The main symptom to suspect bladder cancer is the appearance of blood in the urine (hematuria) without associated pain during urination. The appearance of blood in the urine can be detected with the naked eye (gross hematuria) or in a urinalysis (microscopic hematuria). Although there are several non-tumor processes by which blood can appear in the urine, including urinary tract stones and urinary tract infections, hematuria is an important symptom for which you should always consult with a doctor.
Bladder tumors can also cause urinary discomfort as symptoms, including needing to urinate more frequently and in small amounts, pain or stinging when urinating, or the urge to urinate right after you finish ; and all this without causing associated hematuria. These symptoms are very nonspecific, and may be present in other much more common and benign behavioral pathologies, such as benign prostatic hyperplasia, urinary tract infections or overactive bladder.
When there are metastases in other organs, they can produce very varied symptoms, depending on their location. The most frequent sites where bladder cancer metastases appear are the bones and lung, although they can also appear in the liver, brain and almost any organ.
Diagnosis
When bladder cancer is suspected, in addition to asking about symptoms and exploring the patient, certain complementary tests should be performed to confirm it and to determine the tumor subtype and stage of the disease.
- General blood tests: it is especially important to monitor kidney function, because in patients with bladder cancer it is often altered. Kidney function is determined with a laboratory test called creatinine and glomerular filtration rate. In addition, hematuria can generate anemia or other alterations that will also be visible in this exam.
- Serial urine cytology: The presence of malignant urothelial cells in the urine detected by cytology confirms the diagnosis. However, urine cytology is not very sensitive, and there are usually patients with bladder cancer without notable alterations in their urine analysis, especially in those patients who are diagnosed with low-grade tumors.
- Abdominal and urinary tract ultrasound (reno-vesico-prostatic ultrasound): allows to observe in detail the kidneys, ureters and bladder whenever they are full of urine. It is a non-invasive and harmless test for the patient that in many cases is able to detect bladder tumors, and evaluate their scope on the urinary tract or the rest of abdominal organs.
- Cystoscopy: Cystoscopy is the “gold standard” method for the diagnosis and local staging of bladder cancer. It consists of introducing into the bladder through the urethra a flexible tube equipped with a small video camera. This allows us to observe the inside of the bladder, and even some interventions can be performed, such as removing polyps, taking biopsies, etc. It can be performed in an outpatient operating room, without the need for general anesthesia.
- Computed tomography (CT) scan of the chest and/or magnetic resonance imaging (MRI) of the abdomen and pelvis: usually done especially if the tumor is suspected to have advanced to deeper layers of the bladder. These studies can determine the tumor stage.
In addition, they can be performed in some cases:
- CT or brain MRI: if brain metastases are suspected.
- Bone scan: if bone metastases may exist.
As a diagnostic and treatment technique, it is performed in all cases, with exceptions:
- Transurethral bladder tumor resection (TUR): consists of an operation to be able to observe the inside of the bladder and perform resections in more depth, and thus staging to which layer the tumor has reached. It is an intervention that requires anesthesia, but it is also carried out through the urethra and involves minimal aggression for the patient.
Staging
In general, all tumors are classified into several stages (depending on parameters such as their size, affected organs, distant spread, etc.) that will have a different evolution and require different treatments. In the case of bladder cancer, the most commonly used classification is the TNM system, which assesses the extent of the tumor itself (T, primary tumor), lymph nodes (N) and the presence of metastases (M).
In a simple way, the following stages are distinguished:
- Non-muscle-invasive bladder tumor (NSVT) or “superficial” : these are tumors that do not invade the outer layer of the bladder (muscle layer), although they can invade the inner layers of the bladder (mucosa or submucosa).
- Musculo-invasive bladder tumor (MIVT) or “infiltrating”: The tumor invades the bladder muscle. This fact implies a greater possibility of spreading to other parts of the body, producing metastasis.
- Locally advanced bladder tumor: The tumor invades the tissue surrounding the bladder or invades neighboring organs such as the prostate, uterus, or vagina.
- Metastatic bladder tumor: The tumor is spread affecting the lymph nodes or has distant metastases in other organs of the body.
The stage of each case marks in a very significant way the expected evolution of each patient. However, not all patients with the same stage evolve in the same way and, in the particular course of each case, other prognostic factors, treatments received, patient characteristics, etc. will also be very relevant.
Prognosis
The prognostic factors are characteristics of the tumor and the patient that allow to estimate a priori its subsequent evolution, complementing the stage marked by the TNM system.
In early-stage tumors, an important prognostic factor is tumor grade. A tumor is said to be low-grade when, viewed under a microscope, it closely resembles the tissue from which it originated (in this case, the normal tissue lining the bladder or urothelium); And a higher degree when your cells are so damaged and unstructured that they look almost nothing like the original tissue. Currently, urothelial tumors are divided only between high or low grade. High-grade tumors are more aggressive and have a higher risk of coming back after treatment.
Nor do all patients with tumors in advanced stages have the same evolution. Patients who, when diagnosed, have poorer general condition, anaemia, low blood albumin values and/or visceral distant metastases (especially in the liver) are known to have a worse prognosis and shorter survival can be expected.
Treatment
The treatment of bladder tumors is done depending on the stage they present.
- Non-muscle-invasive (NSVT) or “superficial” bladder tumor:
Patients with “superficial” bladder tumors are cured with appropriate treatment in most cases, although they are at high risk of coming back. Depending on the grade of the tumor and the presence of other factors, treatment may consist only of removing the tumor by cystoscopy or transurethral resection or, in other cases, and administering complementary treatment with immunotherapy or chemotherapy directly into the bladder, through a urinary catheter, through one or more instillations. These intravesical treatments reduce the risk of tumor recurrence and tumor progression from “superficial” to infiltrating
- Musculo-invasive bladder tumor (MIVT) or “infiltrating”:
The main problem of these patients is that they have a relatively high risk of the disease producing distant metastases, and therefore, that this affects their survival. There are several treatment options:
- Radical cystectomy:
involves removing the bladder, organs and nearby lymph nodes. It is the most common treatment. It can be performed open, laparoscopic or robotic. Its biggest drawback is that, when removing the bladder, it is not possible to urinate normally. The most frequently used solutions to reconstruct the urinary tract are:
- Perform a ureteroileostomy or Bricker: It consists of diverting the ureters to a fragment of small intestine (ileum), which will flow into the skin, forming an ostomy. A plastic bag is fixed to this hole (ostomy) by means of an adhesive ring to collect urine and the bag is emptied periodically.
- Perform a neobladder: with tissue from the patient’s own small intestine (ileum) a bag is made to which the ureters are connected. The urine reaches this neobladder located inside the abdomen and can exit to the outside through the patient’s own urethra that is reconnected to the neobladder. It is not always possible to perform this last technique and in some cases it is associated with complications.
In recent years, minimally invasive techniques for the treatment of infiltrating bladder tumor have been standardized. Robotic cystectomy has been shown to present fewer complications in the postoperative period, with less intraoperative bleeding, less transfusion rate, and lower rate of paralytic ileus. Cystectomy with Da Vinci robot, in turn, has allowed a prompt recovery of the patient, allowing a decrease in days of hospital admission, given that postoperative complications are minor, maintaining quality oncological results. These techniques require extensive surgical experience in this area. Therefore, less surgical aggressiveness with robotic surgery, and an expert team, give the best results.
- Conservative bladder preservation treatment: It consists of removing the tumor avoiding removing the bladder, to increase the comfort of the patient. These techniques can only be performed in some patients very selected both by the situation of the disease, and by the situation of the patient himself, and must be carried out by teams with experience and good coordination. When they are performed, a very complete follow-up of the patient must be carried out, to identify possible recurrences of the tumor. There are several alternatives:
- Chemoradiotherapy: Bladder tumors are sensitive to radiation therapy and in some cases can be eliminated with this treatment, which is usually administered simultaneously with chemotherapy.
- Transurethral resection: in rare cases, when tumor invasion is not too deep, it may be an alternative, but it is not standard management.
- Trimodal therapy: It is the most recommended option in the case of opting for bladder preservation. It consists of carrying out a wide TUR that allows to eliminate any tumor remains, and then carrying out the treatment with concomitant chemo-radiotherapy.
- Complementary treatment: as mentioned, one of the main problems of these tumors is that they can reappear, either in the initial area (local recurrence) or in other parts of the body (metastasis). This occurs because despite initial treatment, microscopic disease may persist, which we cannot detect in the tests performed at diagnosis. To try to decrease the risk of the tumor coming back, complementary treatment with chemotherapy may be given. Complementary treatment is not always administered, it depends on the stage of the disease and the characteristics of the patient. The treatment can be done in two ways:
- Adjuvant or adjuvant therapy: The administration of chemotherapy after the tumor is removed. Its ability to decrease the risk of the disease coming back is not absolute, but there is data to support its use. There are also some clinical trials underway to try to increase our knowledge on this topic about the use of systemic immunotherapy with immune checkpoint inhibitors.
- Neoadjuvant or neoadjuvant therapy: The administration of chemotherapy before removing the tumor. The results of some clinical trials indicate that it decreases the risk of the disease recurrence and impacts the survival and long-term evolution of these patients. Its main disadvantage is that it delays the operation. The use of systemic immunotherapy as a neoadjuvant treatment for these patients is obtaining promising results.
- Locally advanced bladder tumor:
Treatment is similar, but is usually more aggressive and usually consists of radical cystectomy, radiation therapy, or chemoradiotherapy. Other less aggressive techniques, such as transurethral resection or partial cystectomy, are not usually used. Complementary treatment with chemotherapy or immunotherapy should also be applied in most cases.
- Metastatic bladder tumor:
The goal of treatment is to control or delay the onset of symptoms and increase patient survival. Treatment with surgery, in addition to being more complicated, does not cure the disease, so it is not usually done except with a palliative intention in very selected cases. Treatment options include:
- Chemotherapy: Traditionally it has been the most frequently used treatment in this phase of the disease although the arrival of new alternatives for these patients is assuming a great change in the clinical management guidelines of these patients. The most active and therefore desirable combinations of chemotherapy drugs are those that contain cisplatin. In general, it is usually administered together with gemcitabine, but is also used in combination with methotrexate, vinblastine and adriamycin, or with gemcitabine plus paclitaxel. However, not all patients are good candidates (“fit”) for treatment with cisplatin. Patients with some degree of renal failure, poor functional status, hearing loss, persistent nerve damage to peripheral nerves and/or heart failure are considered unfit (“unfit”) to be treated with cisplatin. In these cases the chemotherapeutic agents traditionally used have been combinations with carboplatin; although at the present time the use of immunotherapy should be considered if the biological profile of the disease allows it. Vinflunine or paclitaxel are chemotherapy drugs used so far in the second line, and could continue to be an alternative for these patients to consider at some point in the sequence of treatments. However, several studies have shown that systemic immunotherapy with check-point inhibitors is superior to these chemotherapy agents in patients whose first-line treatment with cisplatin has failed.
- Immunotherapy: The arrival of immuno-oncology is already a reality and the use of immune system check-point inhibitors has changed the treatment of bladder cancer. The basis of these treatments is to infuse the patient with drugs that modulate the response of their own immune system against the tumor, so that our own defenses are able to eliminate malignant cells. Five different drugs in this family (pembrolizumab, atezolizumab, nivolumab, durvalumab and avelumab) have already proven to be active, and in many cases superior to classical chemotherapy, for those patients with tumors previously treated with platinum, i.e. in the second-line treatment setting. Also in previously untreated patients, not candidates for cisplatin, and whose tumors express significantly elevated the protein “PD-L1”, the use of immune system check-point inhibitors has proven to be the most appropriate treatment option. Recently, very promising results have been reported from the combination of chemotherapy together with atezolizumab in the first line of treatment, so it is possible that in the near future it could be considered as a reality for patients.
- Targeted therapy:
Urothelial tumors are neoplasms that often harbor multiple underlying genetic alterations. The knowledge of these has allowed us to generate a molecular classification of different subtypes of bladder cancer. On the other hand, these advances have allowed the development of drugs that inhibit specific genetic alterations (such as erdafatinib in the case of mutations in the FGFR gene; or other drugs related to the PTEN or MTOR pathway) whose results in bladder cancer are proving very positive and can probably be used in the short term for the treatment of these patients.
- Immunoconjugates: These are compounds that bind a chemotherapy agent to an antibody specifically directed against a specific tumor target. In this way, chemotherapy against neoplasia is carried out, increasing its activity and avoiding its toxicity by not acting on healthy tissues. Enfortumab-Vedotin is a clear example of a very effective immunoconjugate in bladder cancer, so it is possible that it will be incorporated in the not too distant future to the treatment alternatives currently in force for urothelial cancer.
- Radiotherapy:
it can be used palliatively to treat some symptoms of difficult control such as pain, significant bleeding from the urine, etc …
Tracking
After treatment of bladder cancer it is very important to have regular check-ups. Examinations in patients in whom the bladder is not removed, usually include cystoscopy and serial urine cytology to detect possible relapses in the bladder itself as soon as possible.
In the revisions, in addition to the collection of symptoms and physical examination, an imaging test is usually performed periodically including chest x-ray, abdominal and chest CT or ultrasound of the abdomen and urinary tract.. Depending on the extent of the disease at the time of diagnosis and the patient’s symptoms, different tests may be added.
Prostate cancer
Prostate cancer is a disease that develops mainly in older men. It is usually asymptomatic except in advanced stages of the disease.
Prostate cancer is the most common cancer among men.
Risk factors
- Age: Age is the main risk factor for prostate cancer. The risk of developing prostate cancer begins to increase at age 50 in white men and after age 40 in black men or men with a family history (father or brother) of prostate cancer. Nearly two out of three cases of prostate cancer are detected in men over the age of 65.
- Race: Prostate cancer is more common in black men than in men of other races. In addition, black men are more likely to be diagnosed at an advanced stage, and are more than twice as likely to die from prostate cancer compared to white men. On the other hand, the lowest rate of prostate cancer is seen in Asian individuals.
- Family history: The risk of prostate cancer is strongly influenced by family history. Those men who have a first-degree relative (father or brother) diagnosed with prostate cancer are more likely to develop the disease. Only 5-10% of prostate cancers have a hereditary component. In hereditary prostate cancer the age of cancer onset is earlier (before 55 years) and often patients have first-degree relatives affected by prostate cancer. Genes involved in increased susceptibility to the development of prostate cancer have been discovered.
- Diet: Recent studies suggest that high consumption of animal fats may increase the risk of prostate cancer.
Prevention
Several factors that can prevent the onset of prostate cancer have been studied. None of them, whether dietary supplements, drugs, physical or sexual activity, can currently be recommended as a proven factor in the prevention of prostate cancer.
Symptoms
In the early stages, when the tumor is limited to the prostate, it may be asymptomatic or be accompanied by mild obstructive symptoms attributable to benign hyperplasia (decrease in caliber or interruption of the urine stream; increased frequency of urination, especially at night; difficulty urinating or stinging during urination), since prostate cancer and benign prostatic hypertrophy can coexist.
When tumors are locally advanced they are accompanied by clear obstructive symptoms, in addition there may be hematuria (blood in the urine) or signs of infection (the latter two are rare).
When it comes to advanced tumors, edema or swelling of the legs may appear (due to the growth of regional lymph nodes), bone pain (due to tumor extension to the bone) and even weakness or loss of strength in the legs (compression of the spinal cord).
Diagnosis
Physical examination and complementary tests are used for diagnosis.
- Digital rectal exam: It involves an examination of the rectum in which the doctor inserts a finger into a lubricated glove in the rectum and feels the prostate through the rectal wall for abnormal nodules or areas. The prostate gland is located immediately in front of the rectum, and most cancers begin in the back of the gland, which can be felt during an exam of the rectum. This test is uncomfortable, but it is not painful and takes very little time to perform.
Determination of blood PSA levels (total PSA and PSA ratio): A laboratory test that measures the levels of this marker (Prostate-Specific Protein) in the blood. It is a substance produced specifically by the prostate that can be found in greater amounts in the blood of men who have prostate cancer. However, it should be borne in mind that PSA levels can also be elevated in an infection or inflammation of the prostate such as benign prostatic hyperplasia (enlargement of the prostate of non-cancerous origin). PSA screening, although it has been a controversial topic in the urological scientific world, has shown a benefit by increasing diagnosed cases, decreasing deaths attributed to undiagnosed patients. Patients with PSA 10 ng / ml have 70-80% probability that the disease is localized, if PSA levels range between 10-50 ng / ml 50% will be localized, if the PSA < is > 50 ng / ml only 25% will be localized.
- Nuclear magnetic resonance imaging (MRI): A procedure that uses a magnet, radio waves, and a computer to create a series of detailed pictures of areas inside the body. Due to the latest advances in the PIRADS classification, it has become a fundamental test, useful for diagnosis and necessary for performing prostate biopsies by fusion of images. On the other hand, it is useful in evaluating the extent of the tumor within the pelvis.
Prostatic biopsy
It is usually performed guided by transrectal ultrasound. When cancer is suspected, a small amount of prostate tissue needs to be removed for examination under a microscope. This test will confirm the diagnosis of cancer and will give us an idea of the tumor volume and the degree of aggressiveness (Gleason score).
- Gleason 6: Low-grade tumors
- Gleason 7: Intermediate-grade tumors
- Gleason 8-10: undifferentiated tumors
Image fusion prostate biopsy
Fusion biopsy is a technique that allows to merge in real time the images of the prostate MRI with the transrectal ultrasound, which allows to locate exactly the suspicious nodular area within the prostate and, therefore, better precision in the diagnosis of prostate cancer, by allowing the exact puncture of said suspicious area.
Staging
Without treatment, prostate cancer survival depends on age at diagnosis, overall health, tumor grade, and stage at diagnosis.
Among untreated patients with localized prostate cancer, those with a low Gleason score (6) have a very low risk of dying from their cancer in the next 15 years (4-7%) regardless of the patient’s age at diagnosis; however, those who have an undifferentiated tumor with Gleason 8-10, have a higher chance of dying from their cancer compared to other causes, even if the diagnosis is at advanced ages.
The process used to define whether cancer has spread within the prostate or to other parts of the body is called staging. It is important to know the stage or stage of the disease in order to plan treatment. To rule out bone involvement, bone scintigraphy is performed, and to rule out visceral or adenopathic involvement, axial tomography (CT) is used.
Predictive models
They use PSA, Gleason score, and stage T to predict the likelihood that the patient will be disease-free after local treatment, so patients can be included in 3 prognostic groups:
- Low risk: T1-T2a, Gleason 6 and PSA < 10 ng/ml. More than 85% of patients are disease-free within 5 years of treatment with surgery or radiation therapy.
- Intermediate risk: T2b and/or Gleason 7 and/or PSA 10-20. 50-70% will be free after 5 years.
- High risk: T3 or higher or Gleason 8-10 or PSA > 20. Only 33% will remain disease-free after local treatment.
Treatment
There are three strategies considered standard for the management of localized prostate cancer:
- Surgery
- Radiation therapy with/without hormone therapy
- Active surveillance
The choice of treatment depends on several factors:
- The likelihood that the tumor is limited to the prostate gland and therefore potentially curable.
- The tumor size and histologic grade (degree of aggressiveness of the tumor).
- Age of the patient and general condition, as well as associated diseases.
- Potential side effects of different forms of treatment.
Pre-treatment evaluation
Before choosing the best therapeutic option, it is essential that the extent of the disease is correctly evaluated.
The most important factors to predict the evolution of the disease before treatment are the stage, the level of PSA in the blood, the tumor volume (number of positive biopsies and percentage of the material obtained in the biopsy that is affected by the tumor) and the degree of aggressiveness of the tumor, referred to as the Gleason score.
Surgery: radical prostatectomy (PR)
It consists of completely removing the prostate gland and seminal vesicles. There are 3 ways to do it: openly, laparoscopically or robotically.
DaVinci Robot Prostatectomy is the least invasive and most accurate technique that exists for prostate removal. In this surgery, trocars are introduced into the patient’s abdomen, in which the robotic arms are attached, which the surgeon controls from an integrated external console., The fact that the Da Vinci Robot allows a real three-dimensional vision, associated with the fact that the arms allow all degrees of movement within the patient’s abdomen, are the main advantages of this technique, compared to the open and laparoscopic technique. Robotic Prostatectomy, thanks to these advantages, allows a better immediate postoperative period, as well as a decrease in the rate of urinary incontinence and erectile dysfunction. The fact that it is such a precise procedure allows to preserve the urinary sphincter in an integral way, which significantly decreases the rates of urinary incontinence, as well as to preserve the neurovascular bands, which will cause less impact during the postoperative period in terms of erectile dysfunction or impotence.
In turn, it allows extended lymphadenectomies, that is, removal of lymph nodes, in those cases that are required.
3D Laparoscopic Prostatectomy is a minimally invasive technique that has evolved in recent years. In the same way as in robotic prostatectomy, it allows an excision of the entire gland, although the ability to preserve the structures involved in urinary continence and erectile function is not as precise.
Both techniques allow a good control of the oncological disease.
Currently open radical prostatectomy is in disuse, due to its comorbidities.
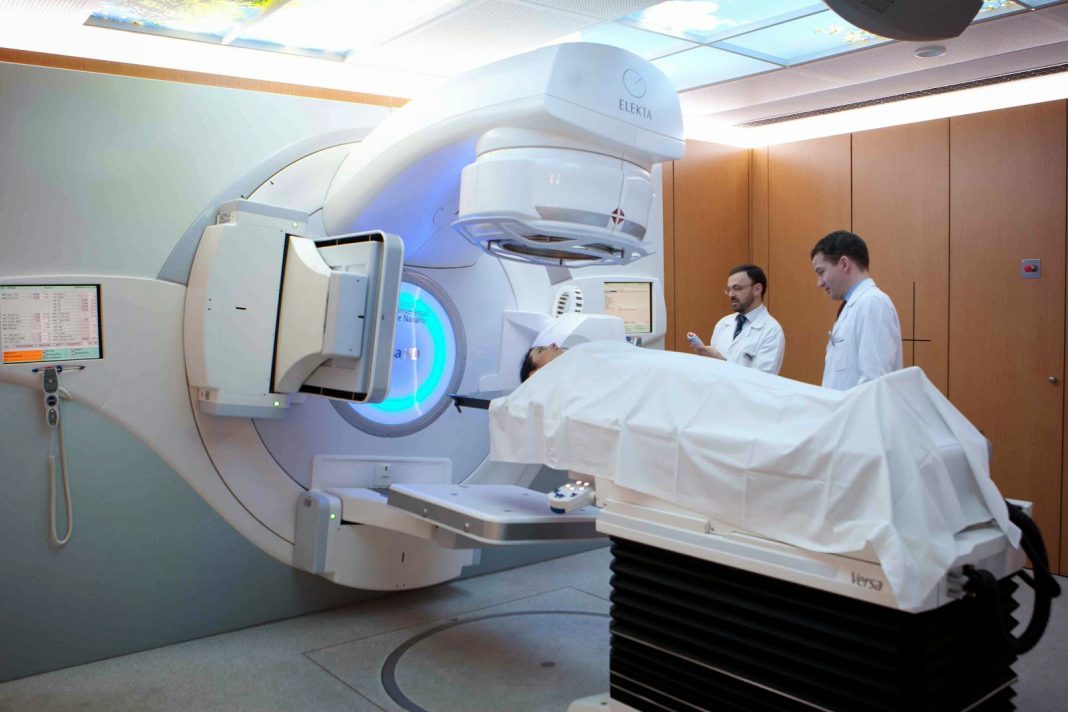
Radiation therapy (RT)
Two forms of RT are used to treat prostate cancer:
- External radiation therapy
- Brachytherapy or RT interstitial implantation
External radiation therapy:
Uses a machine called a linear accelerator that moves around the patient by directing radiation to the pelvis. It is given daily (5 days a week) for 4 to 7 weeks (depending on whether it is used alone or in combination with brachytherapy). It does not require hospitalization. Complications of RT: they are a consequence of the small amount of radiation received by healthy tissues near the tumor (bladder and rectum, especially); with new technological advances they have been minimized more and more. The most frequent are:
- Increased voiding frequency and feeling of voiding urgency
- Painful urination
- Impotence, in this case more frequent as time passes since the RT.
- Intestinal problems (diarrhea, pain and bleeding, the last two secondary to inflammation produced in the rectum called proctitis)
In order to reduce the side effects of classical external radiotherapy, irradiation techniques have been improved and there are now new ways of delivering radiation that are not available in all centers:
- Conformal or three-dimensional RT (3D-RTC): has become standard treatment after the publication of numerous clinical studies. It allows to administer higher doses in the prostate, respecting the tissues that are around, therefore, decreases the rate of side effects, especially intestinal problems.
- Intensity-modulated RT (IMRT): allows the RT dose and intensity to be varied (scaled) during therapy. It reduces side effects and facilitates treatment when pelvic lymph nodes need to be included in the field. It allows to administer very high doses (81 Gy) with little intestinal toxicity. This technique is not available in all centers due to its high cost.
- Brachytherapy or RT of interstitial implantation: consists of administering a radioactive source inside the prostate guided by ultrasound. Advantages: it is done in a short time and requires little hospital stay. The published series seem to indicate that it is a good treatment only for patients with low-risk tumors.
Complications of brachytherapy:
causes an acute urinary syndrome (of short duration) caused by inflammation of the prostate, which can even cause acute retention of urine. It causes less incontinence than external RT, fewer problems at the level of the rectum. The risk of impotence is similar to that of the other treatments.
- Image-guided radiation therapy (IGRT): also allows the administration of higher doses of external radiotherapy (even up to 90Gy) through the use of advanced imaging techniques such as magnetic resonance imaging with spectroscopy.
Active surveillance
It is a valid option in certain cases such as patients presenting with small tumors, with low Gleason, slow rise in PSA levels, and MRI images that do not demonstrate progression. Therefore, it is indicated in those patients who have low-risk tumors
It is not a suitable option for patients with intermediate-risk or high-risk tumors, i.e. patients with large, high-Gleason tumors, in whom tumor growth is rapid and therefore have a high probability of dying from their prostate cancer.
The active surveillance protocol allows to have controlled those patients who have a low-risk prostate cancer, During the follow-up, serial PSA controls are performed, usually every 4-6 months, as well as serial prostate MRIs, usually annual, and prostatic biopsies also serial, annual or biannual, to rule out the progression of the disease. In the event that the disease progresses, active treatment will be carried out.
Approximately 50% of patients who are under observation go on to receive treatment within the first three years either for progression or for the anxiety of remaining untreated.
Hormone treatment: androgen suppression
The prostate is an organ dependent on the level of male hormones, called androgens (the most important of the androgens is testosterone).
Androgenic suppression or deprivation consists of applying treatments that manage to reduce testosterone levels in the body as much as possible, with this it has been observed that the size of both the normal prostate and the tumor prostate decreases.
Studies have shown that adding hormone therapy before or after local therapy (RT) improves outcomes in intermediate- and high-risk patients, but not in low-risk patients.
The duration of treatment is controversial, treatment is usually started 1 or 2 months earlier and extended up to 6 months (intermediate-risk cases) or up to 2 years (high risk) with the data available to date.
Side effects of androgen suppression treatment are: decreased sex drive, impotence, hot flashes, growth of breast tissue that may be painful, muscle weakness, loss of bone mass with increased risk of fractures, anemia, and an increased risk of developing diabetes or coronary heart disease.
Cryotherapy
It is a local treatment that achieves destruction of tumor cells by applying freezing to the prostate gland.
It is applied under local anesthesia in the operating room and although the results are promising, there are no long-term data available, therefore, it cannot be recommended as standard treatment in localized prostate cancer such as surgery or RT. It may be useful in case of recurrence of the tumor after RT or previous surgery.
Focal therapies
Among the alternative treatments that can be applied to patients with low-risk prostate cancer are treatments known as focal therapies. These treatments can be mainly HIFU, although there are also treatments such as photodynamic therapy, electroporation, focal cryotherapy.
Fundamentally, HIFU (High Frequency Ultrasound), allows, after locating the area to be treated based on magnetic resonance imaging and fusion prostate biopsy, to perform a focal treatment, only in the area affected by the tumor. These therapies are currently considered in the research phase, although there is a certain profile of patients who could benefit from it.
Advanced prostate cancer
Approximately 15-20% of patients with prostate cancer present at the time of diagnosis at an advanced stage, that is, the tumor has spread beyond the gland invading neighboring organs by continuity (locally advanced prostate cancer corresponding to tumors T3, T4 of the TNM classification) or affects the regional lymph nodes (stages III) and even invades other organs at a distance (metastatic prostate cancer or stage IV).
On the other hand, patients who in an initial phase present localized tumors, may present recurrence or relapse of their disease, then also being an advanced disease.
Treatment of locally advanced prostate cancer (T3, T4)
Recommended treatment options include:
- External RT with or without brachytherapy
- Transurethral resection (TUR) of the prostate
- Radical prostatectomy
- Hormone therapy based on androgen suppression: usually in combination with surgery and RT.
Surgery
- Radical prostatectomy (RP): As we have said before, it consists of completely removing the prostate gland. They are performed within a modality called “multimodal treatment”. Since the disease is not only located within the prostate, it is proposed to perform after surgery, treatment with RT, or combination of RT plus HT, depending on the characteristics of the tumor after removing the prostate. This allows us to decrease the local recurrence rate. Radical robotic prostatectomy can also be performed in patients with locally advanced prostate cancer
Radiotherapy
- External RT: Another alternative is considered in patients with locally advanced tumors who are treated with external RT in combination with androgen suppression therapy. As for the optimal duration of hormonal treatment there is controversy: it should be started two months before RT and the duration ranges from 6 months to 2-3 years.
- External RT + brachytherapy: as we have seen before, brachytherapy consists of administering a radioactive source into the prostate guided by ultrasound. Its usefulness in locally advanced tumors is in combination with external RT. This option has a duration of 4-5 weeks compared to 8 for external RT but is not recommended in case of very pronounced urinary symptoms.
Androgenic suppression
As we have said before, androgen suppression consists of applying treatments that reduce testosterone levels in the body, with this it has been observed that the size of both the normal prostate and the tumor prostate decreases. It is also a treatment that acts against tumor cells that have left the prostate producing metastasis.
Testosterone levels can be reduced to the maximum in several ways:
- Through a surgery called orchiectomy that involves removing the testicles (where the largest source of androgens in the body is).
- Through drugs that act on the hormone that regulates the amount of androgens produced by the testicles. The most used are: goserelin, leuprorelin, triptorelin, buserelin. All of them are administered by injection.
- Through drugs called antiandrogens: they act on androgen receptors. The most commonly used are flutamide and bicalutamide, which are orally administered drugs. These drugs alone do not sufficiently lower testosterone levels and in general, should be used in combination with the above.
In patients with locally advanced prostate cancer, androgen deprivation is rarely used as the only strategy, since it does not obtain the same results as surgery or RT in terms of local tumor control.
Side effects of hormone treatment: decreased sex drive, impotence, hot flashes, breast tissue growth that may be painful, muscle weakness, bone loss with increased risk of fractures, and an increased risk of developing diabetes or coronary heart disease.
Treatments of patients with PSA elevation after local therapy
When in the follow-up of patients treated for localized and early prostate cancer a sustained elevation of PSA levels in the blood is detected in the absence of symptoms, often with the available diagnostic methods we cannot detect metastases. So we talk about biochemical recurrence. In these cases it is essential to stage well the situation of the tumor, with complementary explorations such as abdominal CT, bone scintigraphy, PET-choline or PET-PSMA, This situation represents a category of advanced prostate cancer in which we must take into account certain considerations:
- Some of these patients may still be curable with additional local therapy.
- The average time it takes for the tumor to show signs of disseminated disease (such as bone metastases) is up to eight years and sometimes there is never symptomatic progression.
However, many patients, although asymptomatic, have high levels of anxiety when they see PSA levels increase progressively.
The best treatment in this situation depends on several factors:
- In patients who have been previously treated with RT, rescue prostatectomy may be considered if the relapse is limited to the prostate and tumor growth is not rapid. The time it takes for PSA levels to multiply x 2 is called the PSA bending time and gives us an idea of tumor growth. Well, when this bending time is less than 3 months, we must consider other options other than surgery.
- Patients initially treated with radical surgery can be successfully rescued with RT. However, local treatment is not recommended in most cases due to the high probability that the tumor has spread beyond the prostate, although we cannot detect it with the available diagnostic methods. In this sense, PET-choline can be useful in an attempt to locate the focus of recurrence.
- For patients not candidates for surgery or RT we have two options: hormone therapy (based on androgen deprivation) or observation.
When to start hormone treatment in biochemical recurrence without symptoms?
The reasons in favor of starting early treatment in the absence of symptoms would be:
- Slow the progression of the disease and decrease the rate of complications such as urinary obstruction and painful metastases.
- There could be a survival benefit, particularly in patients with minimal disease. Not demonstrated in studies to date.
- Many patients do not agree to defer treatment while PSA levels continue to rise with each determination.
The reasons in favor of waiting and starting treatment when there are signs of the disease are:
- Undesirable side effects that clearly reduce the quality of life of the patient and their family environment.
- The high cost of treatment.
The patient should be informed of the benefit / risk of both options and participate with his specialist in the decision to be made, and ideally, after presenting his case in a uro-oncology committee with experts in prostate cancer, both urologists, medical oncologists and radiation oncologists.
Metastatic prostate cancer
First-line treatment
A portion of patients with prostate cancer relapse into advanced disease after local treatment (radiotherapy or radical prostatectomy) and, on the other hand, there is a minority of patients who have disseminated or widespread disease since diagnosis.
When the cancer has spread beyond the prostate gland, androgen suppression is the usually recommended treatment strategy.
There are several options available to reduce or eliminate androgen levels:
- Orchiectomy: Surgery that involves removing the testicles (where the body’s largest source of androgens is).
- LHRH agonist injections: These are drugs that act on the hormone that regulates the amount of androgens produced by the testicles. The most used are: goserelin (Zoladex®), triptorelin (Decapeptyl®), buserelin (Suprefact®), leuprorelin (Procrin®, Eligard®). They are administered by monthly, quarterly or even semi-annual injection.
- Complete androgen blockade: combination of LHRH agonists with antiandrogens that act on androgen receptors, the most commonly used are bicalutamide (Casodex®) and flutamide (Eulexin®). This option leads to greater undesirable side effects, as well as being more expensive. On the other hand, it achieves a modest survival benefit (1-5% at 5 years).
Treatment in androgen monotherapy It is associated with a lower rate of sexual dysfunction and also results in less bone loss. However, it cannot be considered a standard form of treatment for prostate cancer and its use should be restricted to highly selected cases such as low-grade tumors or low-volume metastatic disease.
Intermittent hormone blockade has been studied as an alternative to improve the quality of life of patients requiring hormone therapy. It consists of withdrawing the treatment for a period of time, thus allowing the patient to recover sexual function, at least temporarily. Treatment is usually withdrawn once maximum response is achieved and restarted again when PSA levels rise above a certain level (10-20 ng/ml). However, there is one study that suggests that this strategy is discreetly inferior to the continuous block.
Two large recently reported studies support the role of docetaxel chemotherapy associated with androgen suppression therapy. Both studies conclude that patients with metastatic prostate cancer who receive 6 cycles of docetaxel on a triweekly basis, in addition to therapy with LHRH analogues, achieve survival between 10 and 17 months longer with a good tolerance profile, a magnitude of benefit that had not been observed to date in any solid tumor with the addition of a chemotherapeutic agent. Therefore, chemotherapy with docetaxel should be recommended for patients with metastatic prostate cancer associated with initial treatment with LHRH analogues, especially for patients with “de novo” onset metastatic disease, with large tumor volume (more than 4 foci of bone metastases or presence of metastases in the lung or liver) and provided that they are in good general condition and do not present contraindications for it.
Most patients respond to a first hormonal maneuver, but also virtually all progress to treatment, especially within the first two years. At this stage, they are considered castration-resistant tumors, i.e. androgen suppression alone is ineffective as a treatment. However, patients may respond to other hormonal agents.
Second-line treatment
Virtually all patients with advanced prostate cancer sooner or later develop resistance to androgen suppression hormone therapy, thus progressing to the status of castration-resistant prostate cancer (CRPC) defined as cancer that progresses despite maintaining blood testosterone levels in the castration range (< 50 mg/dl). These patients are candidates for systemic salvage treatment and several options are available today: new hormonal drugs, chemotherapy or bone-directed agents.
Treatments with androgen receptor inhibitors
Abiraterone acetate is an oral drug that prevents the synthesis of androgens both at the level of the testicles, the adrenal gland, and the prostate tumor itself and has been extensively investigated in the treatment of prostate cancer at different stages.
In a randomized study, more than 1,000 patients with metastatic castration-resistant prostate cancer (mCRPC) previously treated with one or two chemotherapy regimens, at least one with docetaxel, patients treated with abiraterone acetate combined with a dose of prednisone or prednisolone had a significant improvement in overall survival compared to patients treated with prednisone or prednisolone plus placebo. Abiraterone acetate treatment achieved a 35% reduction in the risk of death and an increase in median survival (14.8 months versus 10.9 months) compared to placebo-based treatment.
Recently published data also confirm the overall survival benefit of Abiraterone in combination with prednisone in patients with asymptomatic or minimal symptomatic metastatic castration-resistant prostate cancer who have not previously received docetaxel chemotherapy, in addition to achieving an advantage in radiological progression-free survival, a delay in the need for chemotherapy or in the need for opioid analgesics.
Abiraterone has a good tolerance profile, highlighting as more frequent effects fluid retention, hypertension and decreased blood potassium levels; as less frequent effects the drug can raise liver enzymes in the laboratory and could cause heart damage in a minority of patients.
In our country, abiraterone in combination with prednisone is indicated and marketed for the treatment of metastatic castration-resistant prostate cancer in patients who have progressed to docetaxel treatment and in patients who have not received prior chemotherapy, provided they are asymptomatic or have little pain.
Enzalutamide is a new drug from the group of antiandrogens much more potent and effective than bicalutamide, also for oral administration, which unlike abiraterone, does not require concomitant administration of prednisone.
An international multicenter phase III study randomized just over 1,000 patients with metastatic CRPC who had progressed to prior docetaxel treatment, either enzalutamide or placebo, patients treated with the drug had a median survival of 18.4 months compared with 13.6 months for those treated with placebo. This translates to a 37% reduction in the risk of death.
Updated survival data for enzalutamide versus placebo have recently been reported in patients with asymptomatic or symptom-poor metastatic CRPC who have not been treated with prior chemotherapy, confirming a benefit in overall survival, progression-free survival, and time to chemotherapy need.
Enzalutamide also has a good tolerance profile, highlighting as the most frequent effects fatigue, diarrhea that is usually mild and intermittent, muscle pain, headache and high blood pressure.
Enzalutamide is indicated and marketed for the treatment of metastatic castration-resistant prostate cancer in patients who have progressed to docetaxel treatment and in patients who have not received prior chemotherapy, provided they are asymptomatic or have little pain, including in patients who have visceral (liver or lung) metastases.
Both abiraterone and enzalutamide have interactions with other drugs to be taken into account and since the indication of both drugs is for metastatic disease, it is very important to monitor the presence of symptoms associated with the disease, especially the progression of pain. Despite the good tolerance profile, patients receiving these treatments should be closely monitored by their specialist.
Chemotherapy
Chemotherapy was already shown in the 90s effective as a palliative treatment for prostate cancer. The various combinations of cytotoxic drugs studied achieved a biological response (PSA reduction) in more than 50% of patients, with a duration of response of about 6 months.
Two randomized studies in the late 90s demonstrated clinical benefit with a scheme that combined two drugs: mitoxantrone and prednisone compared to prednisone alone, but without improving survival. In 2004, the results of two randomized studies were published that for the first time demonstrated overall survival benefit with chemotherapy regimens containing docetaxel compared to the classic mitoxantrone-prednisone regimen (considered standard until that date).
In both studies, the survival achieved for patients treated with docetaxel reaches 17-18 months, in addition docetaxel reduces the risk of death by 20-24%, significantly increases the PSA response, significantly improves pain and quality of life of patients. The most important side effect of docetaxel is Decrease in the white blood cell count that is the cause of greater susceptibility to infections, other adverse effects that it causes are: hair loss, nausea, diarrhea, fluid retention, alterations in the nails, fatigue and weakness, among others.
The docetaxel-prednisone combination has to this day been considered standard treatment for metastatic castration-resistant prostate cancer.
Until 2010 there was no treatment with proven efficacy in controlled clinical studies for patients in whom docetaxel had failed, the mitoxantrone-prednisone combination was the most used until a few years ago in this group of patients (in the second line of treatment).
A study published in 2010 shows that a new chemotherapeutic agent of the taxane group called cabazitaxel combined with prednisone , achieves a statistically significant improvement in overall survival compared to the classic mitoxantrone-prednisone scheme (15.1 months versus 12.7 months). The most common side effects of this new treatment were diarrhea and the decrease in white blood cell count that causes increased susceptibility to infections. With these results, this drug was the first to be approved in our country for the indication of treatment of castration-resistant prostate cancer that has progressed to chemotherapy with docetaxel.
Bone-directed treatments
Radio-223 is a new agent in the group of radiopharmaceuticals to be administered at the Nuclear Medicine Service. The drug has been shown to improve survival versus placebo (14.9 vs 11.3 months) in symptomatic patients with advanced prostatic carcinoma with bone metastases who had previously received docetaxel chemotherapy or were not eligible for such treatment. Radio-223 achieved a 31% reduction in the risk of death. In addition to this survival benefit, drug administration prolonged the time to the first bone event and added no notable toxicity compared to placebo. It is administered intravenously in 6 doses with a monthly interval between doses. Recently, Radium-223 has obtained the indication in Spain of treatment for castration-resistant prostate cancer with symptomatic bone metastases in patients who do not present visceral metastases.
Treatments that improve quality of life
There are other treatment options available that improve the symptoms associated with the disease and should be applied in combination with tumor-specific treatments (hormone therapy and chemotherapy) to improve patients’ quality of life.
Pain medications or painkillers (from aspirin to opioids) are very effective. As for the possible addiction or dependence with opioid drugs is almost never a problem if they are prescribed in patients with pain of great intensity. Sleepiness and constipation are undesirable side effects , but they can usually be managed by adjusting doses or adding other medications to relieve these effects.
Bisphosphonates are a group of medications that can help relieve bone pain caused when cancer has spread (bone metastases). In addition, bisphosphonates help strengthen bones in patients receiving hormone therapy (prevention of osteoporosis) and are also effective in preventing skeletal complications (pain, pathological fractures, spinal cord compression, and need for surgery or radiation therapy to the bone) in patients with castration-resistant prostate cancer with bone metastases.
The most commonly used bisphosphonate is zoledronic acid, which is administered by intravenous injection of short duration (15 minutes) adjusting the dose to the patient’s renal function.
Bisphosphonates can cause side effects, including flu-like symptoms, bone pain 24 hours after infusion, and a rare but very bothersome side effect is osteonecrosis of the jawbone. (The blood supply to an area in the bone stops and that part of the bone dies.) This can cause tooth loss and infections or open wounds of the jaw bone that do not heal. The only effective way to treat this complication is to stop taking the medication and give drugs to relieve symptoms. It occurs most often after performing dental manipulation such as tooth or tooth extraction while the patient is being treated with bisphosphonates.
Denosumab is a monoclonal antibody directed against a key protein in the progression of bone metastases; it is administered subcutaneously once a month and has an effect on the prevention of skeletal events (pathological fractures, radiotherapy or surgery on the bone and spinal cord compression) associated with bone metastases from different solid tumors, These include castration-resistant prostate cancer.
In a randomized study, denosumab compared to zoledronic acid significantly delayed the onset of the first bone event and reduced both the first and subsequent events.
Both denosumab and zoledronic acid are indicated in the treatment of castration-resistant prostate cancer with bone metastases to prevent bone events associated with metastatic disease; Neither drug has been shown to increase patient survival, however, they do contribute to improving quality of life. There is no scientific evidence to justify the periodic monthly use of zoledronic acid in patients with advanced prostate cancer in the hormone-sensitive phase and it seems that its prolonged use (beyond two years) increases the chances of suffering from osteonecrosis of the jaw.
Denosumab is superior to zoledronic acid in the prevention of bone events with the advantage that it does not need to adjust the dose to the patient’s renal function, although the rate of mandibular osteonecrosis it causes is slightly higher than that of zoledronic acid and at a higher economic cost.
Prevention of osteonecrosis of the jaw:
dental check-up and treat any problems that are detected before starting treatment. Maintain good oral hygiene and undergo regular dental exams.
Kidney cancer
Renal cell cancer (also called kidney cancer) is a disease in which malignant or cancerous cells are found that originate in the lining of the tubules of the kidney (in the renal cortex).
Renal cell cancer is responsible for 2-3% of all malignant tumors in adults. It is twice as common in men as in women. It is more common in African Americans than in Caucasians. Most cases are diagnosed between the 4th and 6th decade of life (from 50 years of age), but can occur at any age.
Risk factors
Renal cell cancer most often occurs sporadically (with no family history) and is rarely part of an inherited syndrome (5% of cases).
The exact causes of this disease have not been determined, however, smoking (doubling the risk of developing this disease), exposure to certain chemicals (cadmium, asbestos, oil), obesity, acquired cystic kidney disease in patients undergoing chronic dialysis (30% of these patients will develop kidney cancer) and indiscriminate use of analgesics. (aspirin and phenacetin, in this case more involved in the development of urinary tract cancer), are associated with an increase in the incidence of this disease.
There are several inherited syndromes that associate kidney cancer of different histological types with other types of tumors. In general, those patients with a first-degree relative diagnosed with kidney cancer before age 40 and those who have bilateral or multifocal kidney cancer in the same kidney may have a hereditary component. It should be remembered that only 5% of kidney tumors that are diagnosed are part of a hereditary syndrome, the vast majority are sporadic.
Classification
Renal cell carcinoma accounts for 80-85% of all renal malignancies. It typically originates in the renal cortex and has several subtypes. Here we describe the most frequent or relevant subtypes:
- Clear cell carcinoma (80%) is the most common. Clear cells can grow from slow (grade 1) to fast (grade 4). In this type of kidney cancer, the results of targeted therapy and immunotherapy have been validated.
- Papillary carcinoma (15%). It is divided into two subclasses: type 1 and type 2 (the latter of more aggressive behavior), are currently treated in the same way as clear cell kidney carcinoma.
- Chromophobic carcinoma and oncocytoma (5%), the latter is very slow growing and rarely has the ability to spread.
- Collecting duct carcinoma (<1%) behaves similarly to transitional cell carcinoma of the bladder and urinary tract.
- Sarcomatoid variant: it can coexist with any of the previous subtypes and leads to a more aggressive behavior of the tumor due to its faster and more invasive local growth. It is called sarcomatoid because when examined under a microscope it resembles sarcoma.
Nephroblastoma or Wilms tumor is another type of kidney cancer that typically occurs in childhood.
Angiomyolipoma is a benign tumor that looks characteristic on computed tomography (CT). It tends to grow and does not usually spread, it can be managed conservatively, embolization or with renal partial surgery.
Symptomatology
Kidney cancer is mostly asymptomatic, so it’s usually a casual finding when performing tests (ultrasound or CT) for other reasons. Many patients do not experience any symptoms until the disease has spread to other organs. . The association of hematuria (blood in the urine), mass and pain in the side has been described as typical, but this triple association is only present in 10% of cases. Hematuria appears in more than half of cases, but may not be visible and may only be evident in a urinalysis (microscopic hematuria).
Anemia, fever and weight loss are relatively common in this type of tumors, when they are in advanced stages of the disease.
Less frequent are polycythemia (increase in the hemoglobin figure) and hypercalcemia (the latter should force us to rule out an extension of the tumor to the bones).
Diagnosis
For the diagnosis, a complete evaluation of the patient is carried out, helping us in complementary tests.
- Physical examination and history: It is important to always examine the patient to check the general state of health and identify any signs of disease, such as masses, lumps or any other signs that seem unusual. The patient is also questioned about their habits, history of diseases and treatments received. It is important to also investigate a history of cancer in the family.
- Blood tests: several analytical profiles are requested that provide guidance on the overall state of health and activity of the disease.
- Urinalysis: test to determine the content of sugar, protein, the presence or absence of blood and bacteria.
- Abdominal ultrasound: useful to identify renal masses and distinguish whether they are cystic or solid in nature.
- Contrast tomography (CT) scan: This is the procedure of choice; this is a procedure in which a series of detailed images of the inside of the body are made from different angles. It is useful for detecting and evaluating renal masses, if there is infiltration of the inferior vena cava, as well as if there is extension to regional lymph nodes.
- Magnetic resonance imaging (MRI): in the event that thrombosis or infiltration of the inferior vena cava is suspected, this technique can help us evaluate the extent of said infiltration.
- Renal arteriography: less used today with the advent of other techniques such as resonance; useful to define the vascularization of the renal tumor and therefore in the evaluation prior to surgery.
- Bone scintigraphy (GGO): to rule out extension to the bones in case of clinical suspicion due to pain or elevation of alkaline phosphatase or calcium levels in the analysis.
- Fine needle biopsy or puncture-aspiration (FNA): The removal of cells or tissues performed so that a pathologist can look at them under a microscope and determine whether or not tumor cells are present. To perform a biopsy of renal cell cancer, a thin needle is inserted into the tumor and a sample of tissue is removed.
- PET-CT: with the latest technology, a PET scan and a CT scan can be done at the same time (PET/CT scan). This allows the radiologist to more anatomically locate the areas of greatest uptake (suggesting an area of cancer) in the PET, although it is not a standard technique and should only be used in selected cases. Its usefulness is greater in other types of cancers.
Staging
The TNM system is used for staging. In summary, the clinical stages based on this system are:
- Stage I: The tumor is 7 centimeters or smaller and is limited to the kidney.
- Stage II: The tumor is larger than 7 centimeters and is limited to the kidney.
- Stage III: The tumor invades the adrenal gland just above the kidney or the layer of fatty tissue surrounding the kidney or major blood vessels in the kidney (such as the vena cava) and may invade the abdominal lymph nodes.
- Stage IV: The cancer has spread to other organs such as the intestines, pancreas, lungs, liver, or bones, causing distant metastases.
Prognosis
The natural history of renal cell cancer is highly variable, and depends on when the diagnosis has been made. Approximately 30% of patients will present as disseminated or metastatic disease at the time of diagnosis and one third of the remaining will develop metastases throughout their evolution.
Prognosis and treatment options basically depend on two factors:
- The stage of the disease: The more stage progresses (from I to IV), the lower the chances of cure.
- The patient’s age and general health.
In advanced or metastatic disease, the levels of hemoglobin, neutrophils, platelets and calcium in the blood, together with general status and time since diagnosis, are factors that classify patients into three prognostic risk groups (Motzer criteria):– —
– Good prognosis group. – Intermediate forecast group. – Poor prognosis group.
Treatment
Different types of treatment are available for patients with renal cell cancer.
Some treatments are standard (the current use treatment), and others are being tested in clinical trials. A treatment clinical trial is a study aimed at helping improve the outcomes of current treatments or to learn about new treatments for cancer patients.
When clinical trials show that a new treatment is better than the standard treatment, the new treatment can become the standard treatment.
| Stage | Treatment |
| Stages I and II | Radical or partial nephrectomy with or without lymphadenectomy Radiofrequency cryoablation or ablation |
| Stage III | Radical nephrectomy with regional lymphadenectomy |
| Stage IV | Cytoreductive nephrectomy Resection of metastases Systemic treatments: • Immunotherapy • Antiangiogenic • mTOR inhibitors • Chemotherapy Palliative radiotherapy Bisphosphonates |
Surgery
When the disease is localized to the kidney, surgery can be potentially curative and should in principle be offered to patients with stages I, II or III.
Surgery that removes part or the entire kidney may be used to treat renal cell cancer. There are the following types of surgery:
- Radical nephrectomy: A surgical procedure that removes the kidney, adrenal gland (currently removed only if infiltration is suspected), surrounding tissue, and usually some nearby lymph nodes. Sometimes part of adjacent organs must also be resected if the tumor is attached. It is usually performed by laparoscopy, robotic surgery or open surgery. It is potentially healing
- Partial nephrectomy: A surgical procedure to remove the tumor and some of the surrounding tissue to preserve healthy, functioning kidney tissue. A partial nephrectomy can be performed to avoid loss of renal function when the other kidney is damaged or has already been removed, also in case of bilateral tumors and should be considered the technique of choice when the tumor is less than 7 cm, since it preserves better renal function. Partial nephrectomy can be performed laparoscopically and robotically. In very rare cases it would be necessary to resort to open surgery.
A person may live with only a part of a functioning kidney, but if both kidneys are removed or are not working, the person will need dialysis (a procedure to clean the blood using a machine outside the body) or a kidney transplant (replacing the diseased kidney with a healthy donated kidney).
Cryoablation and radiofrequency ablation: are other alternatives for local treatment of the tumor if partial nephrectomy is not viable for several reasons (patient’s medical condition). The goal is to destroy the tumor by freezing (cryoablation) or by high energy (radiofrequency). They are useful only in case of small tumors, and patients with significant comorbidities that prevent the surgical procedure.
Surgery in advanced kidney cancer (stage IV): the option of surgery for patients with disseminated or widespread disease aims to reduce the largest possible tumor mass (cytoreductive surgery) and is useful in some patients who are candidates for subsequent treatment with immunotherapy or targeted therapy, especially in those considered as a group of good prognosis and intermediate prognosis. It is not a valid option for patients with multiple metastatic locations and poor general condition.
Metastasis surgery: Metastasis surgery can be useful in very selected cases, especially in small volume and small lung metastases, or also in the case of adrenal metastases.
Minimally invasive kidney surgery (Laparoscopy, retroperitoneoscopy and robotics)
With the intention of minimizing surgical aggression and improving post-surgical recovery, laparoscopic and robotic total or partial nephrectomy techniques were born for the treatment of kidney cancer.
Laparoscopic partial nephrectomy and da Vinci robotic partial nephrectomy have shown less intraoperative bleeding, better recovery of renal function, lower hospital admission, and faster recovery and restart of working life.
In cases where there are previous abdominal surgeries or in cases where the tumor is located on the back of the kidney, the technique of choice is partial nephrectomy by Retroperitoneoscopy, that is, accessing the kidney from the back and side, not from the abdomen. In these cases, recovery is very fast, reduces bleeding and operative time, as well as hospital stay
Radiotherapy
In renal cancer, radiation therapy to the primary tumor is considered a palliative treatment option, that is, non-curative and may be considered in some cases with localized kidney disease and unfavorable medical conditions to undergo surgery. In case of involvement of the surgical resection margins, it could be used as a complementary treatment to surgery, although this strategy does not seem to be clearly demonstrated in clinical studies.
Therefore, in kidney cancer, the role of radiotherapy is reserved to alleviate symptoms derived from bone metastases.
Medical treatment
When it comes to advanced kidney cancer, surgery is not curative and therefore treatment with drugs that act globally and reach all organs of the body (systemic treatment) is recommended in order to achieve remission of the disease or prevent its progression. Recurrent renal cell cancer is cancer that has come back after treatment and can come back in the kidney or other parts of the body, even years after initial treatment. In those cases in which the disease relapses or progresses after surgery, we must also propose a systemic treatment. In selected cases of single relapse, salvage surgery may be considered if feasible.
In the treatment prior to surgery or complementary after it, it should not be considered a systemic treatment to date, in a standardized way.
Chemotherapy
In general, renal cell cancer, unlike other types of tumors, is resistant to chemotherapy. Therefore, we need other therapeutic strategies.
Immunotherapy and cellular check-point inhibitor treatment
It is a type of biological therapy that stimulates the immune system’s ability to fight cancer. Substances produced by the body or drugs made in a laboratory are used to stimulate, direct, or restore the body’s natural defenses against disease.
Several immunotherapy-based strategies have been used to boost antitumor immunity. The administration of classical cytokines as Interleukin 2 (IL-2) and Interferon α (IFNα), has demonstrated clinical efficacy since the 80s, however today its use in the clinic has decreased considerably due to the effectiveness of new target treatments that have shown greater clinical efficacy and better tolerance profile.
On the other hand, new drugs based on inhibitors of cellular receptors “check-point” such as PDL-1 (programmed death 1 checkpoint) and CTLA-4 have appeared. These drugs are Nivolumab, Ipilimumab, Pembrolizumab, Avelumab. When these receptors are blocked, we achieve a sustained immune response against tumor cells.
Targeted therapy
Targeted therapy uses drugs and other substances that can identify and kill specific cancer cells without harming normal cells. These are drugs called target therapies. These drugs can cause remission of the disease or prevent the progression of the disease in most cases but do not cure the disease.
Antiangiogenic therapy It is a type of targeted therapy for advanced renal cell cancer with very consolidated results in recent years. It acts on critical receptors of the blood vessels developed in the tumor itself. With this type of therapy, the formation of new blood vessels by the tumor that are essential for it to be nourished and thus continue advancing is prevented; The result is that the tumor stops growing or shrinks.
Among the antiangiogenic drugs, the best results have been achieved with Sunitinib, Pazopanib, Tivozanib, Bevacizumab, Sorafenib, Axitinib and recently Cabozantinib.
The so-called inhibitors of the mTOR pathway, which is a pathway that has a fundamental role in the metabolism, growth, and proliferation of the tumor cell, have shown activity in this disease in randomized studies and we currently have two drugs: Temsirolimus and Everolimus.
These combinations have demonstrated efficacy in the first line or in the entry treatment for advanced disease:
- Axitinib-Avelumab
- Axitinib-Pembrolizumab
- Nivolumab-Ipilimumab
- Sunitinib
- Pazopanib
- Tivozanib
- Bevacizumab-interferon
- Temsirolimus in patients with poor prognostic criteria
In the second line or after failure to the input treatment:
- Nivolumab
- Cabozantinib
- Axitinib
- Everolimus
- Sorafenib
There are other drugs such as Lenvatinib or other combinations (Cabozantinib-Nivolumab,…) pending in the coming years with promising results.
Symptomatic treatment
In addition to the specific treatment of the disease, the management of symptoms in renal cancer is important. To relieve pain, in addition to analgesic drugs, radiotherapy is useful as a palliative treatment.
In the case of bone metastases, it is also useful to use drugs from the bisphosphonate group (Zometa® -zoledronic acid-) that help reduce the complications derived from tumor involvement in the bone.
Upper urinary tract cancer: renal pelvis and ureter
The ureter is the thin tubular structure that connects the kidney to the bladder. The renal pelvis and ureters are lined with cells of the transitional layer and from these cells most upper urinary tract cancers (75 to 85%) are generated.
It is important to remember that 90% of transitional cell tumors are located in the bladder. Only %-10% is located in the upper urinary tract, so they are also called urothelial carcinoma of the upper urinary tract. Or urinary tract tumor. Those of the renal pelvis double in frequency to those presented in the ureter.
Other histological types are squamous cell carcinoma (15 to 20%), which is associated with urinary lithiasis and recurrent infections, and adenocarcinoma (6-8%).
This cancer is different from kidney cancer.
Primary tumors of the renal pelvis are relatively rare, constituting 7-8% of all renal tumors in humans. Most often occur between the sixth and seventh decades of life and can be benign, but are usually malignant. It is more frequent in men, in a very heterogeneous proportion according to the epidemiological studies that have been carried out: 1.7-4.2:1.
Risk factors
Risk factors for transitional cell cancer of the renal pelvis and ureter include the following:
- Having a personal history of bladder cancer.
- Smoking.
- Chronic use of pain relievers, such as phenacetin (paracetamol).
- Being exposed to certain dyes and chemicals used in the manufacture of leather, textile, plastic, and rubber products.
Symptomatology
The most common symptoms are: hematuria (80-90%), flank pain (24-37%) and bladder irritation (15-20 %).
Routes of dissemination
Cancer can spread by contiguity, lymphatic system, and blood:
- By contiguity. Cancer spreads to nearby areas, such as other areas of the ureter, bladder, adjacent organs (adrenal glands, colon, small intestine), or abdominal wall.
- Lymphatic system. It can spread through the lymphatic vessels and reach the nodes.
- Blood. It can also spread through blood vessels and then settle in any organ of the body.
Diagnosis
Noninvasive and invasive procedures are used in the diagnosis of transitional cancer of the renal pelvis and ureter:
- Physical exam and health history: Your general health is checked and any signs of disease, such as lumps, or anything else that seems abnormal, are identified. Data on health habits, history of illnesses, and previous treatments are also taken.
- Ureteroscopy: The inside of the ureter and renal pelvis is observed and serves to detect suspicious areas that require biopsy. A ureteroscope is inserted through the urethra to reach the bladder, ureter, and renal pelvis. Sometimes an instrument is passed through the ureteroscope to take biopsies or tissue samples and look at them under a microscope for signs of disease.
- Urine cytology: test to check cell characteristics. Sometimes cancer cells are shed from cancers of the kidney, bladder, or ureter that pass into the urine. The positivity range for this type of tumor ranges from 60 to 65% for upper-line tumors.
- Computed tomography (CT) scan: A procedure in which a series of detailed pictures of the inside of the body are taken from different angles. The images are created with a computer connected to an x-ray machine. Iodinated contrast is injected into a vein or swallowed to make organ or tissues stand out more clearly. It has a high diagnostic yield ranging from 78-92% for pelvic and ureter cancer.
- Ultrasound: A procedure in which high-energy sound waves (ultrasound) are bounced off internal tissues or organs to produce echoes. The echoes form a picture of the body’s tissues called an echogram. An ultrasound of the abdomen may be done to help diagnose cancer of the pelvis of the kidney and ureter.
- Magnetic resonance imaging (MRI): A procedure that uses a magnet, radio waves, and a computer to create a series of detailed pictures of areas inside the body, such as the pelvis.
- Biopsy: The removal of cells or tissues for a pathologist to look at under a microscope and check for signs of cancer. This procedure may be done during ureteroscopy or surgery.
Staging
For staging, apart from the diagnostic studies already mentioned, the following may be ordered:
- Positron emission tomography (PET): A procedure to find malignant tumor cells in the body. A small amount of radioactive glucose (sugar) is injected
into a vein. The PET scan rotates around the body and creates a picture of the places in the body that use glucose. Malignant tumor cells look brighter in the picture because they are more active and absorb more glucose than normal cells. Performance is low for this type of tumor cellularity.
- Bone scan: A procedure to check for fast-growing bone cells, such as cancer cells, in the bone. A very small amount of radioactive material is injected into a vein and travels through the bloodstream. Radioactive material collects in bones with cancer and is detected with a scanner. They are performed in those cases in which there is symptomatic suspicion of bone metastasis.
With these diagnostic tests, the disease can be classified within a certain stage:
Stage 0 (noninvasive papillary carcinoma and carcinoma in situ): At this stage, abnormal cells are found in the tissue inside the renal pelvis or ureter. These abnormal cells may become cancerous and spread to nearby normal tissue. Stage 0 is divided into stages 0a and 0is depending on the type of tumor:
- Stage 0a is also called noninvasive papillary carcinoma, which sometimes looks like long, thin lumps that protrude from the tissue inside the renal pelvis or ureter.
- Stage 0is is also called carcinoma in situ; it is a flat tumor in the tissue that lines the inside of the renal pelvis or ureter.
Stage I: Cancer has spread from the tissue lining the inside of the renal pelvis or ureter to the connective tissue layer.
Stage II: Cancer has spread to the muscular layer of the renal pelvis or ureter.
Stage III: Cancer has spread:
- from the muscular layer of the renal pelvis to the fat surrounding the renal pelvis or to the kidney tissue; or
- from the muscular layer of the ureter to the fat surrounding the ureter.
Stage IV: Cancer has spread to at least one of the following:
- a nearby organ.
- the layer of fat surrounding the kidney.
- lymph nodes.
- to other parts of the body, such as the lung, liver, or bone.
Stages 0, I, and II occur in most diagnosed patients (55 to 65%) with a 5-year survival ranging from 87% to 100%. Some researchers find that 5-year survival for stages III and IV ranges from 12% to 50%.
In addition, cancer of the renal pelvis and ureter may be classified as localized, regional, metastatic, or recurrent:
- Localized: the cancer is found only in the kidney.
- Regional: cancer has spread to tissues around the kidney, nearby lymph nodes, and blood vessels in the pelvis.
- Metastatic: cancer has spread to other parts of the body.
- Recurrent: the cancer has recurred (come back) after treatment. Cancer may come back in the renal pelvis, urethra, or other parts of the body, such as the lungs, liver, or bones.
Treatment
Treatment options depend on the following:
- The stage and grade of the tumor.
- Where the tumor is.
- Whether the patient’s other kidney is healthy.
- Whether the cancer has recurred.
Less and less invasive treatment protocols are being carried out, with the aim of preserving the renal unit and offering a better quality of life to patients. This is thanks to the advent of important technological advances in endourology and the exhaustive training of our specialists.
Standard treatment
Organ sparing endourological procedures (Endoscopic laser treatment of upper urinary tract tumor)
Laser flare: It is a surgical procedure that destroys tissue using a laser beam. The surgery is carried out by ureterorrhenoscopy or percutaneous access, that is, using thin instruments that are inserted through the natural holes or through a small incision in one side. Not all tumor stages and grades can be treated this way; It is reserved for low-grade tumors, single, smaller than 2cm and that do not produce obstruction of the urinary tract. Laser fulguration can be considered a curative treatment only in specific situations, since between 50-70% of recurrences are recorded after the procedure, which would require a very close follow-up and repeat every so often the exploration of the urinary tract. The great advantage of this procedure is that it allows the preservation of the kidney, avoiding its removal. It is indicated in patients with low aggressive urothelial tumors in patients with only one kidney or patients with renal failure to avoid dialysis.
Surgery
One of the following surgical procedures is used to treat transitional cell cancer of the renal pelvis and ureter:
- Nephroureterectomy: Surgery to remove the entire kidney, ureter, and bladder cuff (tissuethat connects the ureter to the bladder). It is indicated in localized tumors that are not amenable to endoscopic management with laser flare. It is also performed in locally advanced or metastatic disease, representing a palliative option. The surgery is completed with a regional lymphadenectomy (removal of lymph nodes in the pelvis).
- Segmental resection of the ureter: A surgical procedure to remove the part of the ureter that contains the cancer and some of the healthy tissue around it. Then, the ends of the ureter are reattached. This treatment is used when the cancer is superficial and is located only in the lower third of the ureter, near the bladder. Performing this procedure in other ureteral locations is associated with a high rate of recurrence. The surgery is completed with a regional lymphadenectomy (removal of lymph nodes in the pelvis).
In any of these techniques, minimally invasive surgery can be used, either laparoscopic surgery or robotic surgery. Laparoscopic and robotic radical nephroureterectomy have shown good oncological results, as well as a lower rate of intraoperative bleeding and a better postoperative
Chemotherapy and cellular check-point inhibitor treatments
Adjuvant chemotherapy (after surgery) achieves a recurrence-free rate of up to 50%, and has an effect on survival. Not all patients receive this treatment due to concomitant diseases and impaired renal function after radical surgery. The treatment regimen used is based on the use of cisplatin, in combination with other chemotherapy drugs. In cases where the disease progresses despite the use of chemotherapy, or in the event that renal function is not good and, therefore, chemotherapy cannot be administered, treatments based on cellular check-point inhibitor treatments such as Pembrolizumab, Atezolizumab and Nivolumab may be used.
Radiotherapy
The use of postoperative radiotherapy for malignancies of the renal pelvis remains controversial, as they are assumed to be radioresistant. There is little evidence for its use in upper line tumors.
Prognosis
Prognosis depends on stage and grade of the tumor. The 5-year survival for transitional cell carcinoma is 50%, but if the tumor is undifferentiated only 25% of patients survive after 5 years. Squamous cell carcinoma has an unusually aggressive ability to spread to regional lymph nodes, and therefore any type of treatment does not modify its survival, being almost always fatal within a year and with a five-year survival of less than 10%.
Because of the multicentric nature of these tumors and the propensity for recurrence, patients should be carefully monitored even after radical surgery.
Early diagnosis and timely treatment of these neoplasms are fundamental factors in the prognosis of these patients.
Tracking
Cystoscopy, urinary cytology, and CT scans of the chest, abdomen, and pelvis are performed every 3 months for 2 years, every six months for the next 2 years, and annually thereafter. The reason for performing cystoscopy so often is because 30 to 50% of patients with transitional cell carcinoma of the renal pelvis can develop this neoplasm synchronously in the bladder.
For tumors treated by laser fulguration, ureterorenoscopies are performed every 3 months for 2 years, and then every 6 months, although it is true that protocols may vary depending on the operative findings and the patient’s health status.
Benign tumors of the adrenal gland
The adrenal glands are small organs that are located, as the name implies, above and adjacent to both kidneys. They are shaped like an inverted Y, V or T and weigh approximately 5 grams each. The adrenal cortex secretes cortisol, aldosterone, and androgens; The marrow secretes epinephrine and norepinephrine.
Benign tumors of the adrenal gland are common, affecting 9% of the population; however, cancers are very rare. Tumor detection has increased significantly with the increasingly frequent use of computed tomography (CT) and magnetic resonance imaging (MRI), in which many of these lesions are detected incidentally.
The most common lesions are adenoma and metastases, with adenoma being the most prevalent pathology in patients with no neoplastic history. In addition to these, at the adrenal level you can find pheochromocytomas, carcinomas, lymphomas, myelolipomas, cysts and pseudocysts, hemangiomas and hematomas, among others.
Adenomas
They are a common finding in CT and MRI. They are small, non-functioning, found in asymptomatic patients and correspond to an incidental finding. A minority of adenomas are hyperfunctioning, responsible for endocrine disorders such as Cushing’s syndromes (excess glucocorticoids) or Conn syndrome (hyperaldosteronism), in which adrenal adenomas represent 20% and 80% of these, respectively. This lesion is characterized histologically by the presence of clear cells with abundant intracytoplasmic lipid (microscopic fat) separated by fibrovascular tissue.
Metastasis
It is the most common malignant lesion of the adrenal glands, this being the fourth most frequent site of metastasis in the body, despite the size of these glands. Primary neoplasms that frequently metastasize to the adrenal glands come from the lung, breast, kidney, intestine, and pancreas. Melanoma is a rare tumor that also metastasizes to these organs. Metastases are frequently bilateral, larger than adenomas, heterogeneous and with irregular contours in imaging tests.
Pheochromocytoma
It is a rare, catecholamine-secreting tumor originating from the neuroectodermal tissue of the adrenal medulla. Symptoms of this tumor are secondary to excess epinephrine and norepinephrine, including episodic high blood pressure, headache, anxiety, and palpitations.
It has been called the tumor of the 10%s: 10% bilateral, 10% extraadrenal, 10% malignant, 10% in children, 10% associated with syndromes, such as multiple endocrine neoplasms NA and III, Von Hippel Lindau syndrome and neurofibromatosis. The diagnosis is clinical and laboratory (elevation of plasma and urinary catecholamine) and on the other hand through complementary tests, either by CT or MRI, which present a similar performance in the detection of these tumors in the adrenal glands.
Pheochromocytomas are usually between 2 and 5 cm long, hypervascular and have plenty of fluid, many with cystic areas inside. When they are located in the extraadrenal situation, they are called paragangliomas and caudal should be sought at the origin of the inferior mesenteric artery or adjacent to the aortic bifurcation.
Radical surgery is the only treatment that can lead to complete remission.
Myelolipoma
A rare benign tumor composed of mature and hematopoietic adipose tissue. Most are discovered incidentally; Sometimes they may bleed.
Lymphoma
Involvement by this disease is rare, being in most cases the non-Hodgkin type. Of the patients with non-Hodgkin’s lymphoma, 4% have adrenal involvement.
Cystic lesions
They are rare, being demonstrated in 0.1% of autopsies. They are usually asymptomatic, and occur preferably between the third and fifth decade of life. They can be associated with pain or palpable mass when they are very large. They are divided into four subtypes: endothelium-lined cysts (40%), posthemorrhage pseudocysts (39%), parasitic cysts, epithelium-lined cysts.
Bruises
They occur in relation to trauma, especially when there is involvement of the right hepatic lobe and direct compression of the ipsilateral adrenal gland against the spine. They can also be visualized against the background of adrenal vein thrombosis, hypotension and shock.
Surgical treatment of benign tumors of the adrenal gland
Laparoscopic surgery in adrenal adenomas was the first laparoscopic surgery to demonstrate the current advantages of minimally invasive surgery. Laparoscopic surgery, retroperitoneoscopy surgery and robotic surgery of the adrenal gland have shown less operative bleeding, shorter operative time, better postoperative recovery, shorter hospital stay and shorter time until recovery from work activity, so they are currently considered the techniques of choice for adrenal surgery.
Malignant tumor of the adrenal gland (adrenal carcinoma)
Rare tumor (0.02% of all cancers, with overall incidence between 0.5-2/1,000,000 inhabitants), originating from the adrenal cortex, usually between the fourth and seventh decades of life. It affects both sexes, with a slight female preponderance, especially towards the end of the third and fourth decades, although it can occur at extreme ages. It is hyperfunctioning in 20-40% of cases, manifesting more commonly as Cushing’s syndrome. It may present with abdominal pain and palpable mass.
At diagnosis, the tumor is large, usually larger than 6 cm, reaching in some cases up to 20 cm. Direct invasion of neighboring organs, renal vein and inferior vena cava may be observed. Approximately 50-60% of cases have clear evidence of locoregional or distant dissemination at the time of diagnosis.
The etiology is unknown, and no predisposing factors have been identified; However, tumor cells are known to display various enzymatic failures in steroid genesis systems (11-beta-hydroxylase, 17-alpha-hydroxylase, 21-hydroxylase, 3-beta-hydroxysteroid dehydrogenase, and delta5-delta4-isomerase). Therefore, it is assumed that the tumor origin takes place at some point in this enzyme failure. In addition, a loss of alleles has been demonstrated on chromosomes 11p, 13q and 17p of carcinomas not present in cases of adenomas or hyperplasias.
Adrenal tumors are more common within the spectrum of several genetic syndromes, such as Gardner syndrome, Li-Fraumeni syndrome and Beckwith-Wiedemann syndrome. Also, 52% of tumors show immunohistochemical positivity for the p53 gene, 80% for the retinoblastoma (RB) gene, 22% for the neuroblastoma gene, and 20% for the von Hippel Lindau gene.
Symptoms
The tumor is palpated as an abdominal mass in 30-40% of patients as a result of its slow growth. Depending on the size and location, it can produce gastrointestinal symptoms such as early satiety, feeling of bloating, nausea and vomiting. Asthenia, fatigue, weight loss, and low-grade fever are common symptoms, as is abdominal pain. The tendency to invade vascular structures is the cause of atypical symptoms secondary to inferior cava thrombosis (peripheral edema), renal vein (hematuria and proteinuria) or portal trunk (portal hypertension with hepatosplenomegaly).
Functioning tumors can produce substances that cause a wide variety of symptoms. In men, the symptoms are included in a clinical syndrome called hyperandrogenism (increased hair thickness, temporary baldness, acne). In the case of women, 75% of these tumors manifest themselves in the form of virilization (hirsutism, amenorrhea, clitoral hypertrophy, appearance of beard, temporary entries and baldness). These symptoms are mediated by the secretion of the hormone DHEA-S. In both sexes hypercortisolism can occur, developing a Cushing’s syndrome (full moon face, buffalo morrillo, plethora, ecchymosis, hypertension and hypokalemia with equal frequency, skin thinning, stretch marks and left ventricular hypertrophy). In boys, it manifests with precocious puberty. Less frequently, symptoms may occur due to hypersecretion of mineralocorticoids, insulin, erythropoietin, vasopressin, estrogen and growth hormone.
Metastasis: in terms of distant spread, adrenal carcinoma metastasizes in the lung (60%), liver (50%), lymphatic (48%), bone (24%), pleura and heart (1%), with metastasis to kidney (5%) and brain (4%) being very rare.
Clinical-histological classification
It can be divided into:
- Differentiated variant, composed of cells that resemble normal ones, arranged in an organized way.
- Undifferentiated or pleomorphic variant, composed of cells with bizarre nuclei, disordered, with lack of cohesion with each other.
Diagnosis
When adrenal carcinoma is suspected, the diagnostic sequence is aimed at demonstrating the degree of function, identifying the degree of local invasion and ruling out the presence of metastasis.
Radiological study
The first imaging technique to be used in the patient in whom adrenal carcinoma is suspected is CT or MRI, followed by I-cholesterol scintigraphy that provides additional information about the functional nature. Positron emission tomography (PET) with deoxyglucose or fludeoxyglucose-F-18 has proven to be a great method for diagnosing adrenal masses in patients with known carcinomas.
Hormonal study
In addition to radiological tests, functional studies are used, in which attempts are made to detect metabolic residues of the substances produced by the tumor itself, due to its own enzymatic failures (the most frequent are that of 11-beta-hydroxylase and 3-beta-hydroxysteroid dehydrogenase). This study is called chromatographic analysis of the various steroids. The alterations found are:
- Those produced by Cushing’s syndrome: marked elimination of cortisol in blood and urine
- Those produced by hyperandrogenism: marked elimination of 17-ketos and 11-deoxycortisol in urine. Increased plasma DHEA-S. DHEA-S is the best marker of carcinoma and its plasma concentrations remain elevated and not suppressed to normal values after suppression with 2 mg dexamethasone/day for 5 days.
- Those produced by feminization / precocious puberty: elevation of estradiol, estrone and / or estriol; increased urinary elimination of 17-ketos in urine.
- Those produced byldosteronism: elevation of precursors such as DOCA and 18-hydroxycorticosterone. In addition, hypokalemia appears. Lack of response to ACTH infusion is typical. Also, carcinomas do not show the classic decrease in aldosterone due to postural changes.
Staging
The disease can be classified, depending on its extent, into:
- Localized disease: tumor that after its removal is confined to the adrenal and cannot reveal the existence of metastasis. Less than 5% of adrenal carcinomas fall into this category.
- Regional disease: Tumor with involvement of adjacent tissue(s) and neighboring organs, including lymph nodes, but no evidence of distant metastasis. Between 20-30% of tumors are included in this group.
- Distant disease: tumors metastasized at diagnosis. This group includes 40-60% of tumors.
- Recurrent disease: occurrence of local recurrence or distant spread after theoretically curative resection of the primary tumor.
Depending on the stadium, it can be classified into:
- Stage I: (T1N0M0). Tumor smaller than 5 cm without nodes, without local invasion and without metastasis.
- Stage II: (T2N0M0). Tumor larger than 5 cm without nodes, without local invasion and without metastasis.
- Stage III: (T1 or T2N1M0 or T3N0M0). Any size, with nodes or local invasion, without metastasis.
- Stage IV: (any T, any NM1, or T3-T4N1). Any size, with lymph nodes or local invasion, with metastasis.
Prognosis and evolution
The prognosis depends on several histological features displayed by the tumor: presence of extensive areas of necrosis, fibrosis bands, capsular invasion, vascular invasion, tumor architecture (organized or diffuse), proportion of clear cells, nuclear pleomorphism, mitotic activity and presence of atypical mitosis.
In general, the prognosis is very poor, with mortality between 65-94% at 5 years, a median survival of 29 months and a median actuarial survival of 35% at 5 years. By stage, 5-year actuarial survival can be estimated at 54-46 for stages I-II, 43-21% for stage III and %-46% % for stage IV.
Treatment
Although adrenal carcinoma is a tumor of high degree of malignancy and a short life expectancy, a treatment should be tried that at least allows an acceptable time of remission, and in cases of functioning tumor, better control of symptoms.
Surgery
Surgery, with curative or palliative overtones, is indicated in cases of tumors with local or regional disease, while in patients with disseminated tumors the indication for palliative cytoreduction surgery may be debatable, and may be elective again in cases of isolated single metastases in affordable organs and in cases of some local recurrences. On other occasions, debulking surgery is indicated to facilitate the action of adrenolytic or chemotherapy drugs.
The approach is open in cases of involvement or suspicion of involvement of neighboring organs. Laparoscopy is, in this case, reserved for lesions located within the gland. In a high percentage of cases it is necessary to perform a block nephrectomy. In addition, a regional lymphadenectomy is performed, which fulfills a double purpose: oncological and staging.
In selected cases with invasion of the vena cava, thrombectomy or resection of the infrarenal cava may be indicated, although the results are so hopeless that it does not seem to be a routinely advisable maneuver.
Adrenolytic treatment
Postoperative treatment with some adrenolytic-acting drugs may improve and prolong survival when initiated immediately after surgery, although it is not known whether this treatment can prevent the occurrence of local recurrences.
The drug of choice is mitotane or Lysodren (at doses of 1-2 g/day up to 5-6 g/day), used for the treatment of metastatic disease. This medication improves symptoms due to hypercortisolism because, by blocking 11-beta-hydroxylase, it drastically reduces cortisol synthesis. In addition, it helps to reduce the possible occurrence of local recurrences and is currently the only possible alternative in the disease with extensive spread at a distance. Studies with this drug have confirmed measurable tumor remissions of 34-60% and overall survival increased by 20-25%. In any case, the degree of clinical response does not exceed 25%. The side effects of mitotane are: nausea (88%), vomiting (23%), diarrhea (38%), weakness, dermatitis (23%), gynecomastia (50%), arthralgias (19%), leukopenia (7%), hypercholesterolemia, hepatotoxicity, neurotoxic symptoms (loss of memory and ability to concentrate in 50%, drowsiness and dizziness) and prolongation of bleeding time due to an effect similar to that of aspirin. The response, if it occurs, has an average duration of 6-10 months.
Chemotherapy
Cortical carcinoma is very insensitive to chemotherapy drugs and has been claimed to be an intrinsically resistant tumor to chemotherapy.
Radiotherapy
Radiotherapy has only a palliative role, especially in painful bone metastases.
Adrenal carcinomas in children
In children, as in adults, adrenal cancers are usually tumors larger than 4 cm, with a male preponderance of 2/1 and an average age of presentation of about 5 years. The most common form of presentation is hirsutism (50%) and virilization (76%), Cushing’s syndrome (30%) and feminization (10%), adrenal tumor not functioning (8%) or very rarely associated with a syndrome of saline loss due to congenital adrenal hyperplasia. Associations with malformations of other organs, such as hemihypertrophy, Beckwith-Wiedemann syndrome, vascular malformations, urological or brain tumors, are not uncommon. 57% have palpable tumor, 80% local disease, 7% locoregional disease and 13% distant metastases at diagnosis. As in adults, the median survival is approximately 50% at 5 years.
Pathology of the urachus
The urachus or middle umbilical ligament is a midline structure that extends from the anterior aspect of the bladder to the navel. It is a remnant of embryonic development that comes from the allanthoid (yolk sac). It is normal for it to be obliterated by 15 weeks of gestation, and to be a fibrous cord by 20 weeks.
The persistence of urachus can cause several clinical problems, not only in children, but also in adults. The diseases that affect it are rare (1 in 5,000 people) and their abdominal manifestation, symptoms or urinary signs are nonspecific, so their diagnosis is not always easy.
The classic presentation triad of urachus pathology is:
- Secretion through the navel.
- Abdominal pain.
- Urinary symptoms.
Depending on the mode of presentation, the pathology of the urachus can be:
- Persistent urachus. When the fibrous cord that extends from the navel to the bladder dome remains permeable (open). May contain narrow, anfractuous or dilated areas. It is more common in men than women, with a 2:1 ratio. Persistent urachus may be associated with congenital anomalies of the lower urinary tract, such as posterior urethral leaflets or complete ureteral atresia. They can be complicated by infections and, in very rare cases, by cancer. Unfortunately, urachal cancer has a poor prognosis, since at the time of diagnosis they usually present local invasion.
- Urachal diverticulum. It develops when the supra-vesical portion of the urachus does not close and is usually asymptomatic. On imaging tests it appears as a cystic-looking profile protrusion, in the midline. It is complicated by urinary tract infections, formation of intraurachal stones and is related to an increased prevalence of carcinoma.
- Urachal sinus. It occurs when the most dilated portion of the persistent urachus is the one closest to the navel, continuing with anfractuous tracts as it approaches the bladder. It can manifest with fetid secretions through the navel, cellulite, stinging or pain in the wall of the abdomen and formation of qualifications inside the breast.
- The urachal cyst. It develops when the urachus is obliterated at the bladder and umbilical ends, remaining between these points a permeable portion that accumulates secretions, taking on a cystic aspect. This usually occurs in the lower third. Their size is variable, but they are usually small. When they become symptomatic they increase their size with reinforcement of their wall. Its diagnosis can be by ultrasound or computed tomography, in which it manifests as a fluid-filled cavity in the midline of the lower abdominal wall. As in other urachal abnormalities, infection is the most common complication.
Diagnosis
The diagnosis is made by physical examination, palpating the abdomen and detecting the presence of the dilated tract under the skin or calcifications in the urachal tract. Sometimes, this is not easy, and less in obese patients.
Imaging tests that are commonly performed include ultrasound, tomography, and magnetic resonance imaging. They are very useful tools, because they allow diagnosing the type of urachal pathology and the possibility of associated malignancy.
Cystoscopy also assists in diagnosis, as it allows the scanner to directly observe the communication of the persistent urachus with the dome of the bladder.
Treatment
Patients diagnosed with a sinus infection, diverticulum, or urachus cyst are prescribed antibiotics with prolonged regimens and local grooming.
The vast majority of the time the definitive treatment is surgical. Surgery can be laparoscopic or open, and includes excision of the entire urachal cord, bladder dome, and sometimes the navel.
- ADRENALECTOMY
- LAPAROSCOPIC RADICAL NEPHRECTOMY
- LAPAROSCOPIC PARTIAL NEPHRECTOMY
- ROBOTIC PARTIAL NEPHRECTOMY
- LAPAROSCOPIC NEFROURETERECTOMY
- ROBOTIC NEFROURETERECTOMY
- TRANSURETHRAL BLADDER RESECTION
- LAPAROSCOPIC RADICAL CYSTECTOMY
- OPEN RADICAL CYSTECTOMY
- ROBOTIC RADICAL CYSTECTOMY
- RECONSTRUCTION OF THE URINARY TRACT (SHUNT OR NEOBLADDER).
- BRICKER URETEROILEOSTOMY.
- ILEAL NEOBLADDER
- LAPAROSCOPIC RADICAL PROSTATECTOMY
- ROBOTIC RADICAL PROSTATECTOMY
- PRESERVATION OF NEUROVASCULATRES BANDELETAS
- LAPAROSCOPIC/ROBOTIC INGUINAL/PELVIC/RETROPERITONEAL LYMPHADENECTOMY
- URETHRECTOMY
- PARTIAL PENDECTOMY
- TOTAL PENDECTOMY
- RADICAL PENDECTOMY
- INGUINAL ORCHIECTOMY